Benzynes, also known as Arynes, are highly reactive intermediates formed in organic chemistry aromatic reactions.
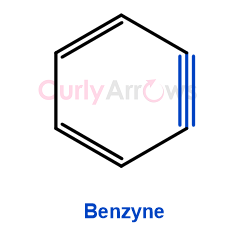
Benzynes are identified as containing a triple bond between two adjacent carbons of the benzene ring. This triple-bonded Benzyne is generated using very strong bases under harsh reaction conditions.
They are formed due to the elimination of two adjacent substituents from a benzene ring and forming an additional bond, leaving behind a highly strained and reactive intermediate.
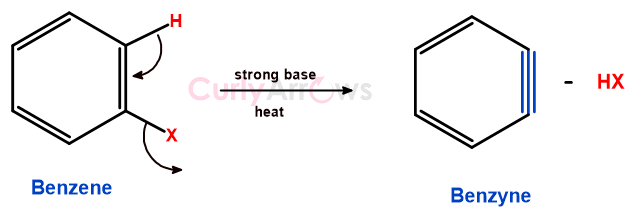
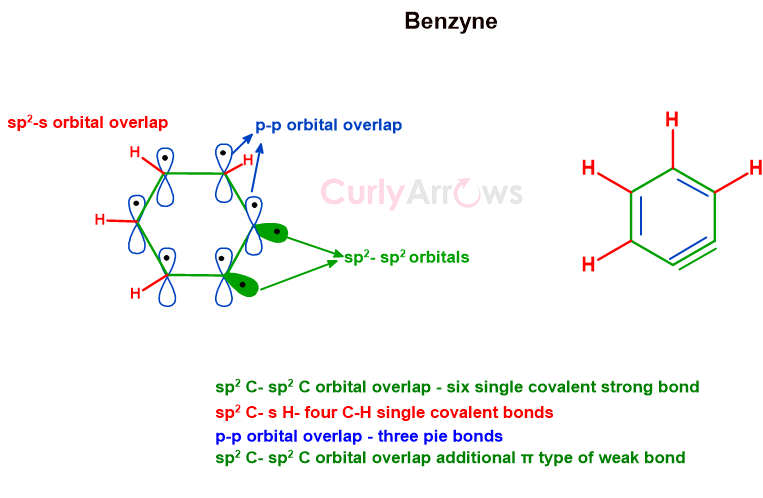
The difference between the additional bond of Benzyne versus benzene is that the lateral p-orbital overlap forms benzenes’ pie bond, whereas the lateral overlap of two sp2 orbitals of carbon forms benzynes’ bond.
Unlike the head-on overlap of the sp2-sp2 orbitals in C-C sigma bonds or sp2-s in C-H bonds, this type of side-by-side sp2-sp2 orbital overlap produces a weaker, highly strained intermediate that can break easily.
This benzyne intermediate can be stabilized to a certain extent by having electron-withdrawing groups on the benzene ring that takes away the electron excess due to the additional bond, thereby decreasing the strain.
Such highly reactive Benzyne is not isolated but generated in situ (that is, within a reaction) to undergo nucleophilic additions, cycloadditions, and polymerization reactions at the triple bond upon which the stability of the benzene ring is regained.
