Heterolytic cleavage or heterolysis is a chemical reaction in which the bond between two atoms breaks unequally so that the two bond electrons reside with only one atom.
The atom that receives both electrons become an electron-rich negatively charged ion (anion, denoted with a negative sign), and the atom that lost the electrons forms a positively charged ion (cation, denoted with a positive sign).
For example, a heterolytic cleavage of tert-butyl chloride, as shown below, generates two species- a cation (electrophiles) and anion (nucleophiles or leaving group).
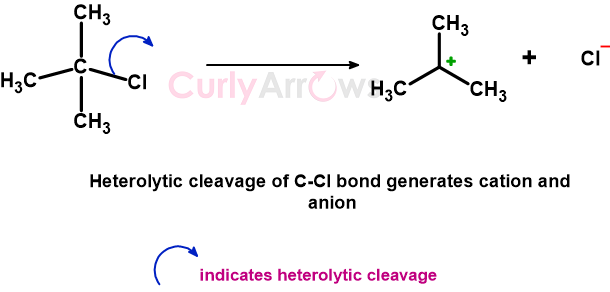
Heterolytic cleavage is indicated using a full-headed curly arrow. The arrow base is on the bond, and the arrowhead is on the atom that receives both the bond electrons.
This type of bond cleavage is often seen in molecules containing polar covalent bonds, where one atom has a greater electronegativity or the ability to pull the bond electrons more than the other. Such a bond cleavage results in the electronegative atom attaining a negative charge while the other atom becomes positively charged.
Heterolytic bond cleavages are common in organic reactions like substitution, elimination, addition reactions, etc.
Related Reading: Nucleophiles, Electrophiles, Leaving Groups.
