Once the sigma bonds are formed, any additional electrons in the perpendicular unhybridized p-atomic orbitals engage in side-to-side or lateral overlap to form an additional bond known as the pie bond. The symbol π denotes the pie bond, drawn as an additional line over the sigma bond in the molecular structure.
The engagement of two electrons from two p-orbitals of each atom forms one pie bond, resulting in a double bond (=). Similarly, the overlap of two more electrons from additional p-orbitals gives two pie bonds, and with the previous sigma bond, a triple bond (≡) is formed.
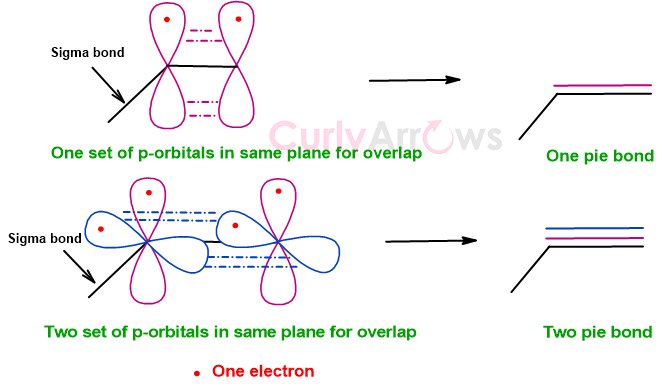
As the pie bond holds two atoms in additional orbital overlap, bridging their distance and reducing the bond length, their bond rotation is also restricted.
The pie bond is weaker than the sigma bond since the electron densities are diffused and placed on either side of the internuclear axis resulting in a less efficient overlap. Due to this, it’s easier to break a pie bond than a sigma bond.
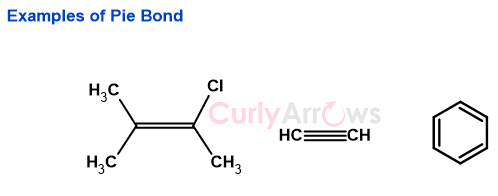
Examples-