The single covalent bond is referred to as a sigma bond, denoted by the symbol σ.
The sigma bonds are usually mentioned in valence bond theory to visualize the bond formation between atoms to form polyatomic molecules like H2, CH4, etc., by overlapping their atomic orbitals.
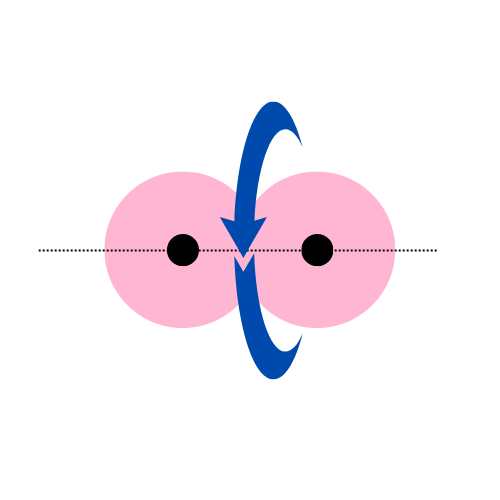
When two similar or dissimilar atomic orbitals approach along the central line or axis of the nucleus, known as the internuclear axis, it leads to a direct head-to-head overlap of the atomic orbitals creating the strongest, most efficient single covalent or the sigma bond.
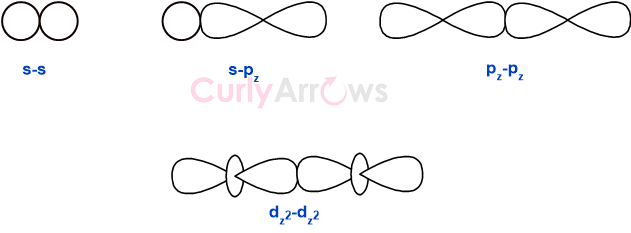
For example, the head-on-head overlap of the two s-orbitals and two p-orbitals along the internuclear axis is shown below.


Since the head-to-head overlap occurs along the internuclear region, it has a larger overlapping area.
Also, once the bond formation completes, the bond undergoes symmetric rotation along the internuclear axis.
Common orbital overlaps forming a sigma type of covalent bond are- s+s, pz+pz, s+pz, and dz2+dz2 (where z is defined as the axis of the bond or the internuclear axis).