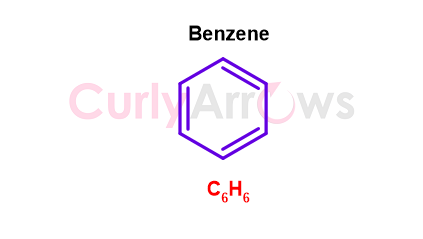
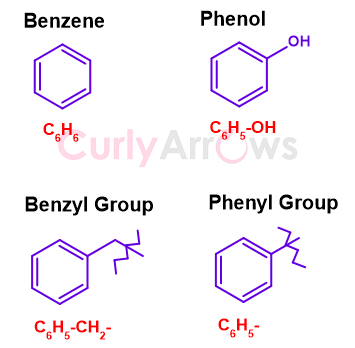
It is easy to identify the simplest aromatic hydrocarbon Benzene that is made of only two atoms- Carbon and hydrogen. Benzene is a planar, cyclic ring molecule with six sides. It has an alternating double bond that contributes to its extra stability by endowing it with resonance, a phenomenon of electron delocalization. The molecular formula of Benzene is C6H6 and the structure is-
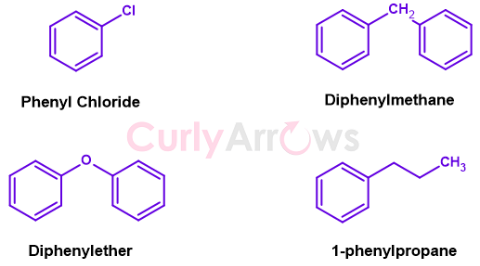
Removing one hydrogen of the Benzene gives a phenyl (Ph-) group, written as C6H5-. If the lost hydrogen is replaced with Chloro, it becomes phenyl chloride (or chlorobenzene). On using phenyl as a side chain for propane, we get 1-phenylpropane. Replacement of two hydrogens of methane with phenyl group gives diphenylmethane.
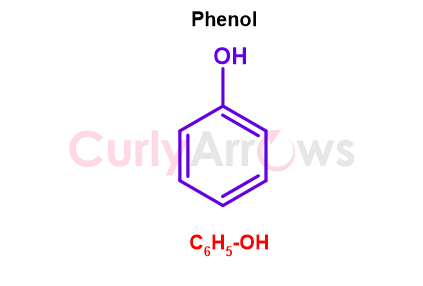
When one hydrogen of the benzene ring is replaced with a hydroxy/alcohol functional group (-OH), the molecule obtained is a called phenol (combination of phenyl and alcohol). Therefore, phenol is the hydroxy derivative of benzene having the molecular formula C6H5-OH.
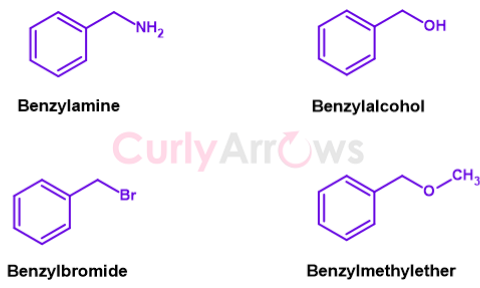
Adding one-Carbon linker (methylene, -CH2-) to the phenyl gives a benzyl group (C6H5-CH2- or Ph-CH2-) abbreviated as Bn. The methylene can be monosubstituted (C6H5-CHR-) or disubstituted (C6H5-CR2-).
Bn= C6H5-CH2- or Ph-CH2-
If the functional group present at the end of the alkyl chain is an amine, the molecule is called benzylamine (Bn-NH2). Other examples of different functional groups are benzyl alcohol (Bn-OH), benzyl bromide (Bn-Br), benzyl methyl ether (Bn-O-CH3), etc.
All the molecules encountered in the early examples are Benzene derivatives. However, for other non-benzene molecules displaying aromaticity (similar properties of planarity and resonance), in that case, such a molecule is simply referred to as an Aryl and is written as an Ar-.
The aryl group can be benzene, substituted benzenes, benzene with heteroatoms of N, S, O, naphthalene, other heteroaromatic compounds of thienyl, indolyl, etc. A few examples of aryl groups are-
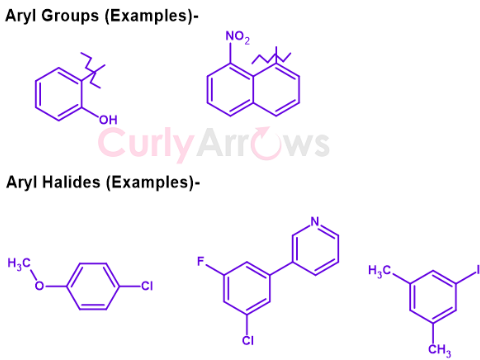
Shown below are all the groups in one hypothetical structure-
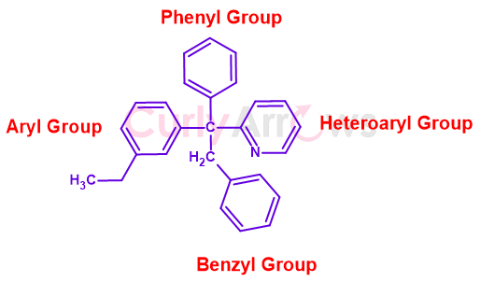
.png)