The nomenclature is mainly used for sp3 hybridized Carbon of an alkane. The Carbon of alkyl halides, alcohols, carbocations, and the Nitrogen of amides and amines are classified by this method.
A primary, secondary, tertiary, and quaternary Carbon and Nitrogen are denoted by numerals and with a degree (o) sign in the superscript. Primary Carbon and Nitrogen are indicated by the number 1, whereas secondary, tertiary, and quaternary are expressed using numbers 2, 3, and 4. Therefore, the representations are – the primary (1o), secondary (2o), tertiary (3o), quaternary (4o).
A Carbon is called primary if it has one neighboring alkyl carbon atom. Secondary Carbon has two, and tertiary has three such linked alkyl carbon atoms.
For 3-methyl pentane (shown below), the terminal methyl are primary (1o) since it has only one Carbon neighbor. The penultimate ones are secondary (2o), and the central one is tertiary (3o) Carbon.
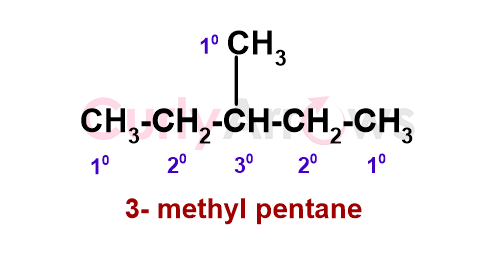
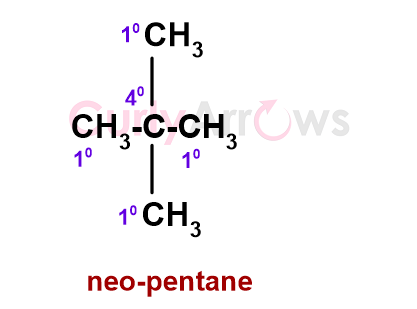
Some alkanes have a quaternary (4o) Carbon, that is, a Carbon linked to four other alkyl carbon atoms - for example, neo-pentane.
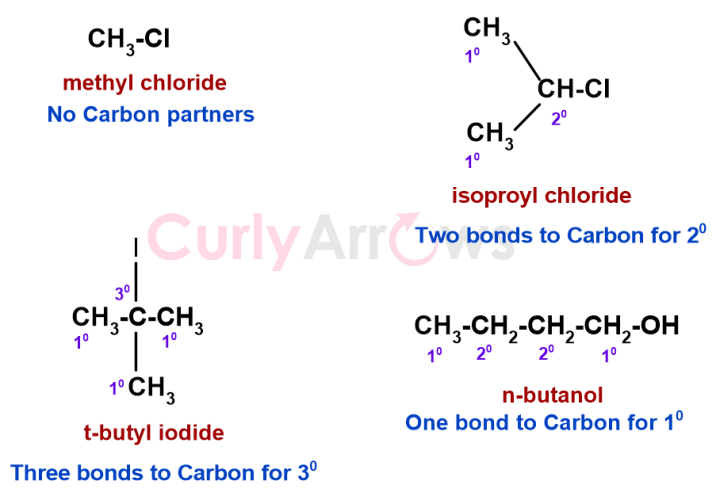
According to this naming convention, for the Carbon atoms part of alkyl halides and alcohols, the functional group is not counted as a Carbon neighbour.
In the examples below, the methyl in methyl Chloride has no Carbon partners and therefore is not a primary carbon atom. The Carbon next to the functional group (-OH) in n-butanol has one immediate Carbon partner, so it is a primary (10) carbon atom.
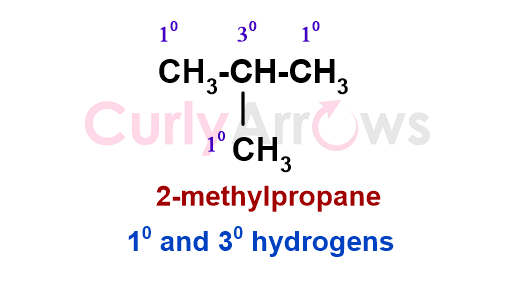
The hydrogens attached to a 1o, 2o, 3o carbon are called 1o, 2o, and 3o hydrogens.
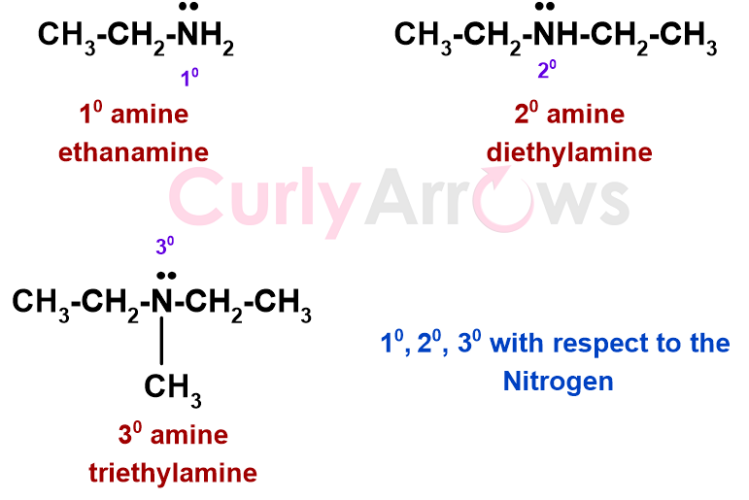
The number of carbons attached to the nitrogen significantly contributes to an amine or an amide. A primary amine has one carbon linked to the nitrogen (for example, CH3-CH2-NH2), a secondary amine has two (for example, CH3-CH2-NH-CH2-CH3), and a tertiary amine has three (for example, (CH3-CH2-)3-N).
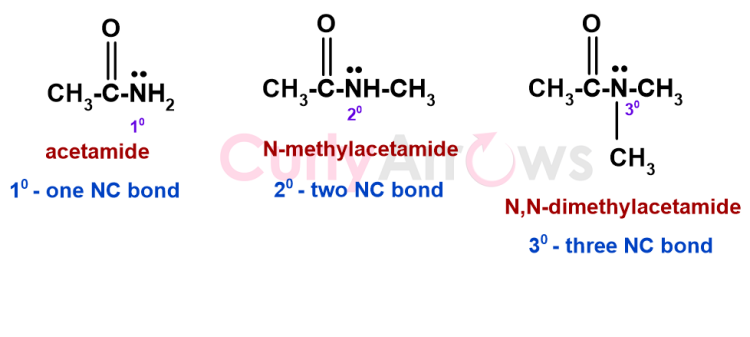
The same convention is applies to the amides. A 1o amide would have only a Carbon neighbor. For example, in CH3-CO-NH2, the -NH2 group is linked to the carbonyl carbon and is, therefore, a 10 amide. A secondary amide would have two carbon neighbors (CH3-CO-NH-CH3), and a tertiary amide would have three. For example, CH3-CO-N(CH3)2.
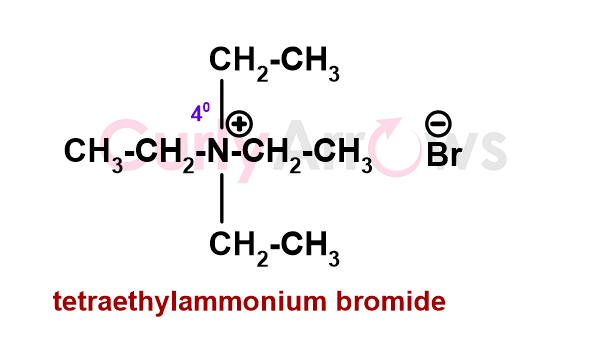
In amines, if the Nitrogen lone pair participates in bonding, it forms an additional bond. Such a Nitrogen atom is called ammonium due to the positive charge on the Nitrogen. It primarily exists as salt, and the Nitrogen is called quaternary (40). The compounds formed are called quaternary ammonium derivate (R)4N+ that exist alongside a negative counterion (for example, Cl-, HSO4- etc.). An example of a quaternary ammonium salt is tetraethylammonium bromide.
The amide Nitrogen does not participate in the quaternary salt formation and, therefore, is not found.
Alkyl carbon next to sp2 carbon
10, 20, 30 Carbon naming is not used when the alkyl carbon is next to sp2 hybridized carbon of an alkene or the carbonyl functional group.
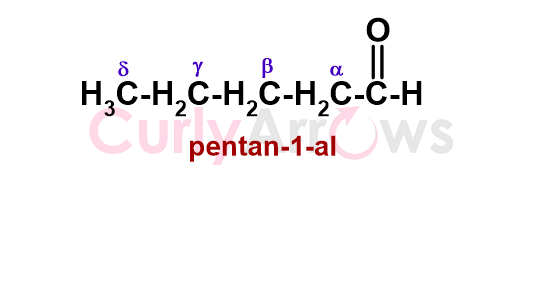
The chain of sp3 carbon atoms neighbouring the carbonyl carbon is denoted using Greek alphabets of alpha (α), beta (β), gamma (γ), delta (δ). The alpha carbon is the closest and the delta farthest to the carbonyl functional group.
The terms primary (10), secondary (20), tertiary (30), and quaternary (40) do not apply to a carbon involved in the pie bond. For example, carbonyl carbon, phenyl carbon, alkenyl carbon, alkynyl carbon.
Aim of the Nomenclature
The terminology explains trends in a substrate's reactivity and stability under certain reaction conditions. For example -
The order of reactivity of alkyl halides towards SN1 is- -CH3 < 1o < 20 < 30
The order of the acidic strength of alcohol is - 1o> 2o > 3o
The stability of the carbocation follows the order- 3o> 2o> 1o
The basicity of the amines in the gaseous phase is - 3o> 2o> 1o
Take the quiz to identify primary, secondary, tertiary and quaternary Carbon and Nitrogen.
