Question- A and B are two functional isomers of compound C3H6O. On heating with NaOH and I2, Isomer B forms a yellow precipitate of Iodoform whereas Isomer A does not form any precipitate. Write the formulae for A and B.
1) Functional Isomers are compounds that have same molecular formula but different functional groups.
At first, the degree of unsaturation (double bond or ring) in the compound having the general molecular formula C3H6O must be found out.
Degree of unsaturation= (2C + 2 + N - H - X)/2
where,
C- Nos. of Carbon Atoms
N- Nos. of Nitrogen Atoms
H- Nos. of Hydrogen Atoms
X- Nos. of Halogen Atoms
= (2 x 3 +2 -6)/2 = 1 (indicating the presence of one double bond or a ring)
Note that in the calculation, the divalent Oxygen is not part of the equation.
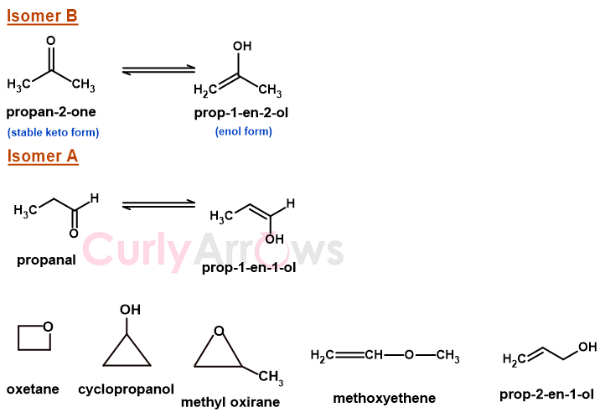
2) A positive Iodoform test (yellow precipitate on treatment with NaOH/I2) confirms the presence of a methyl ketone CH3-CO-. Therefore, the Isomer B is CH3-CO-CH3 (propan-2-one). You can also write it in the tautomeric enol form as Prop-1-en-2-ol.
3) Isomer A gives negative Iodoform test. The probable structures of isomer A that satisfies both the condition can be-
i) Propanal (CH3-CH2-CHO) or its enol form Prop-1-en-1-ol
ii) Oxetane
iii) Methyl Oxirane
iv) Cyclopropanol
v) Methoxyethene
vi) Prop-2-en-1-ol