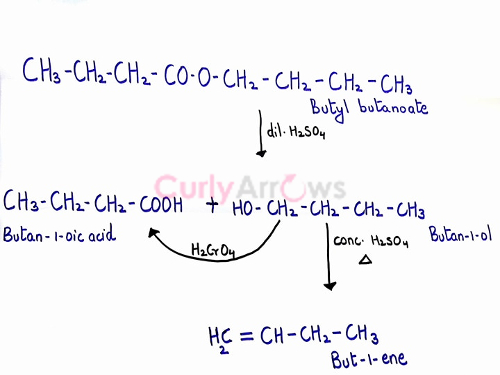
An Organic Compound 'A' molecular formula C8H16O2 was hydrolyzed with dilute H2SO4 to give a carboxylic acid 'B' and an alcohol 'C.' Oxidation of 'C' with chromic acid also produced 'B.' On dehydration 'C' gives 1-but-ene. Write the equations for the reaction involved.
1) The first step is find out the Degree of Unsaturation (the number of double bonds or the presence of a ring) in the compound.
The formula is,
Degree of Unsaturation= (2C + 2 + N - H - X)/2
where,
C- Nos. of Carbon Atoms
N- Nos. of Nitrogen Atoms
H- Nos. of Hydrogen Atoms
X- Nos. of Halogen Atoms
Note that divalent Oxygen and Sulfur atoms are not part of the equation.
Therefore, Degree of Unsaturation= (2x 8 + 2 - 16)/2= (18-16)/2= 1
There is a presence of one double bond or a ring (cycloalkane) in the compound.
2) The first statement mentions acidic hydrolysis of a compound 'A' that generates an acid and an alcohol.
An 'ester' on acidic hydrolysis gives an acid and an alcohol molecule. An ester has one carbonyl carbon that satisfies the number of double bonds required as per the formula.
3) Statement two mentions the oxidation of alcohol 'C' produced 'B' implying the same number of carbon atoms in both the molecules. So on equal distribution of eight Carbon atoms to give an acid and alcohol, we get,
CH3-CH2-CH2-COOH (butan-1-oic acid) and CH3-CH2-CH2-CH2-OH (butan-1-ol). The butan-1-ol on oxidation with chromic acid would give butan-1-oic acid.
4) On dehydration, butan-1-ol will lose water molecule to give but-1-ene.
Therefore, the correct structure of the starting ester is CH3-CH2-CH2-CO-O-CH2-CH2-CH2-CH3.
The reaction steps are,