1) For comparison of the relative oxidizing and reducing powers of various metals:
A Positive number indicates stronger reduction potential of the electrode, and it functions as a stronger oxidizing agent. A negative number means weaker reduction potential of the electrode, and hence it is a more powerful reducing agent.
Example,
The F2 electrode with the standard reduction potential of + 2.87 Volts is a strong oxidizing agent and Li+ ion with the standard reduction potential value of -3.05 V is a strong reducing agent.
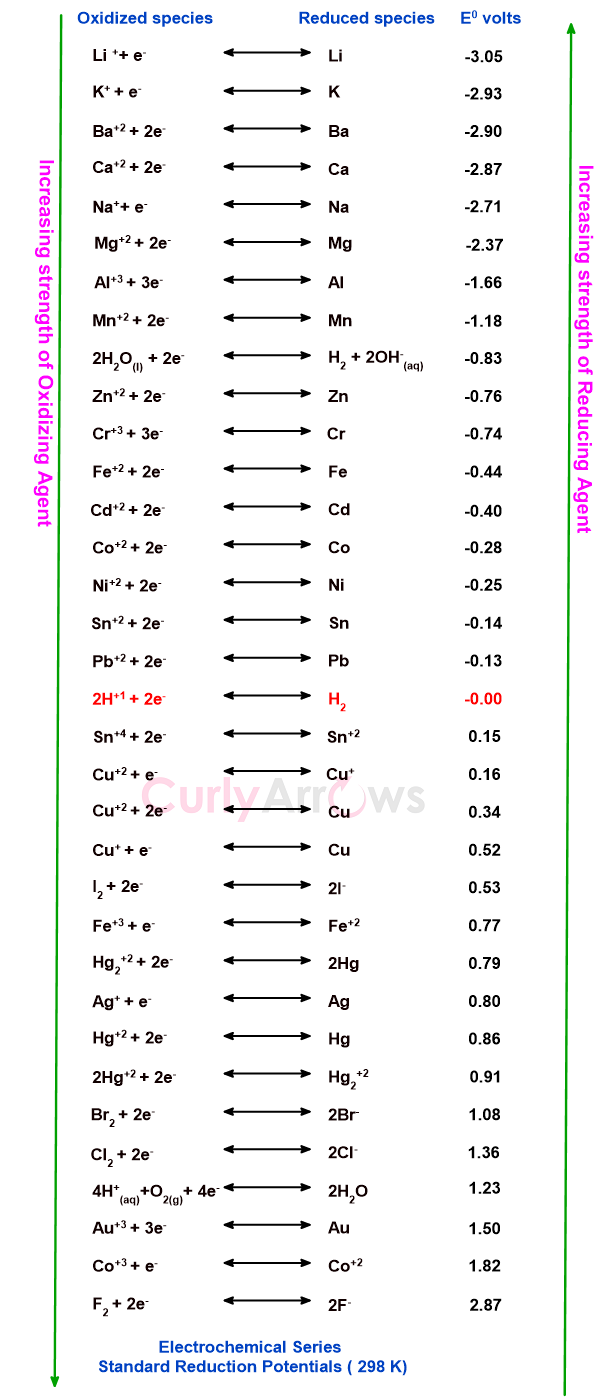
2) For comparison of the relative activities of metals towards displacement reactions based on their placement in the activity series:
As all the metal displacement reactions are cation displacement reactions , the rule of single displacement theory is applicable here.
The single displacement theory says-
‘The element that forms cations can displace only cations and the elements that form the anions can displace only the anions.'
Greater the oxidation potential of metal, more quickly can it undergo loss of electrons and greater is its reactivity. Thus, a metal with greater oxidation potential can displace metals with lower oxidation potential from their salt solution.
In other words, any metal that is higher in the activity series can displace other metal that is placed lower in the activity series, from its compound.
Example 1,
2Al + Fe2O3 → Al2O3 + Fe
Al (-1.66 V) has higher oxidation potential than Fe (- 0.44 V) and displaces Fe to form Al2O3.
Example 2,
Mg + CuSO4 → MgSO4 + Cu
Mg with an oxidation potential of -2.37 V displaces Cu (0.34 V) from CuSO4 to give MgSO4.
3) To calculate the Standard EMF of any electrochemical cell (Eocell):
The standard EMF of the cell is an addition product of standard electrode potential of two half-cells: Reduction half-cell and Oxidation half-cell.
E0cell = Eored + Eoox
By convention, for an electrochemical cell, the standard oxidation potential is always expressed in terms of the reduction potential.
Thus, the standard oxidation potential (Eoox )= - Standard reduction potential (Eored).
Therefore,
E0cell = (Standard Reduction Potential of the reduction half reaction) - (Standard Reduction Potential of oxidation half reaction)
As the anode is the negative terminal for oxidation and cathode for reduction, hence,
E0cell = Eocathode - Eoanode
Example 1:
For a reaction,
Zn(s) + Pb2+(aq) → Zn2+(aq) + Pb(s)
The Standard reduction potentials given are: Pb2+(aq)│Pb(s) is -0.125 V and Zn2+(aq)│Zn(s) is -0.764 V.
We must calculate the Standard EMF.
From the reaction, we can see that Zn loses the electrons and Pb2+ gains the electrons hence, oxidation half-cell or the anode is the Zn electrode.
Using the formula,
E0cell = Eocathode - Eoanode
E0cell = -0.125-(-0.764) = -0.125 + 0.764 = 0.639 V
It is important to note that if standard electrode potential is expressed as oxidation potential, then remember to change the sign before using it in the equation as is shown in the next example.
Example 2,
Zn(s) + 2Ag+(aq) → Zn2+(aq) + 2Ag(s)
Given: E0 Zn(s),Zn2+(aq) = 0.65V and E0 Ag(s),Ag+(aq) = - 0.75 V.
When expressed in terms of the reduction potential, the change in value is as,
E0 Zn2+(aq),Zn(s) = - 0.65V and E0 Ag+(aq),Ag(s) = + 0.75 V.
By looking at the reaction, we can conclude that the oxidation half-reaction takes place at the Zn electrode. Hence it is acting as the anode.
E0cell = Eocathode - Eoanode
E0cell = 0.75-(-0.65) = 1.4 V
4) In predicting the feasibility/occurrence of a redox reaction:
For a redox reaction to be spontaneous or occur readily, the EMF of the cell must be positive. If it comes out to be negative, then the direct, spontaneous reaction cannot take place.
Such information is important in predicting the outcome or the stability of a metal salt solution when stored in another metal container.
Example,
Can a solution of 1M Copper Sulfate be stored in a nickel vessel?
Given is: E0 Ni2+(aq),Ni(s) = -0.25V and E0 Cu2+(aq),Cu(s) = +0.35 V.
Ni + CuSO4 → NiSO4 + Cu
We want to see whether Ni metal will displace copper from CuSO4 solution to give NiSO4 by undergoing oxidation reaction.
Ni(solid) + Cu2+(aqueous) → Ni2+(aqueous) + Cu(solid)
As seen from the reaction, the oxidation terminal will be the Nickel electrode.
Using the formula,
E0cell = Eocathode - Eoanode
E0cell = 0.35 - (-0.25) = 0.60 V
As the EMF come out to be positive, it implies that copper sulfate will react when placed in a Ni vessel and hence cannot be stored in it.
5) To Predict whether a metal reacts with acid to liberate Hydrogen gas:
For a metal to react with acid (H+), the following reaction should take place,
M(s) + H+(aq) → M+(aq) + ½ H2(g)
The metal undergoes oxidation, and the hydrogen ion by accepting the electron undergoes reduction. - If the metal has negative reduction potential and is placed above the Hydrogen in the electrochemical series, it can react with an acid to liberate hydrogen gas.
Lower is the reduction potential (more negative), higher is the reactivity of the metal with the acid.
Example 1,
2Na + 2HCl → 2NaCl + H2 ↑
Sodium metal (- 2.71 V) reacts with Hydrochloric Acid to liberate Hydrogen gas.
Example 2,
Will a Zn metal strip when placed in a 1M H2SO4 solution react with it to liberate H2 gas?
Given: E0 Zn2+(aq),Zn(s) = -0.76 V
For the reaction to occur, Zn would undergo oxidation, acting as the anode terminal.
The cell representation is:
Zn│Zn2+ ║H+│H2
As we know, the standard reduction potential of the hydrogen electrode is taken as zero.
Putting these values in the formula,
E0cell = Eocathode - Eoanode
E0cell = 0 – (-0.76)= 0.76 V
the EMF of the cell obtained is a positive number implying that the reaction is favored.
The metals copper, silver, gold, and platinum and placed below the hydrogen are less reactive, and they do not replace hydrogen from water or acid.
