Pre-requisite Reading- Valency of Elements, Modern Electronic configuration, atomic orbitals, Concept of Hybridization
sp3 hybridization of Carbon
The three common hybridization states are - sp3, sp2, and sp. The sp3 (pronunciation: ess-pee-three) hybridization of Carbon explains its four bonds’ tetravalency, shape, and equivalency.
Carbon (atomic number Z=6) in an unbonded state (ground state) has an electronic configuration of 1s2 2s2 2px1 2py1. The electrons in the 1s atomic orbital are called the core electrons. The core electrons are closest to the nucleus, experiencing maximum attractive force, and therefore do not participate in any bonding reactions. The electrons in the 2s and 2p atomic orbitals are called the valence electrons, and they participate in bond formation reactions.

The 2s and 2p atomic orbitals are separated by an energy gap, with the 2s orbital being lower in energy than the 2p atomic orbitals. This energy gap is overcome when Carbon can form bonds with other atoms, mainly atoms of the p-block (Carbon, Hydrogen, Halogens, Oxygen, Nitrogen, etc.). The process excites one of the two electrons in the 2s atomic orbital of the ground state carbon atom to jump to the higher energy empty 2pz orbital.
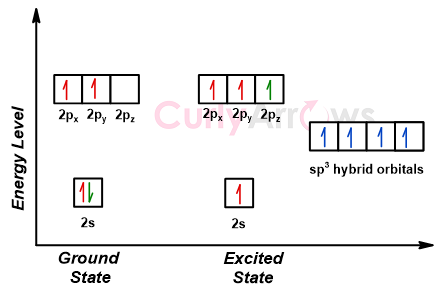
The excitation results in mixing of 2s and the 2p atomic orbitals to form a new set of four hybrid orbitals with more unique identities. These four hybrid orbitals do not have an energy gap seen in the parents- 2s and 2p atomic orbitals and are equivalent in energy. Their energy lies in the middle of 2s and 2p but is a little closer to 2p than the 2s atomic orbitals. The shape of the new hybrid orbital is like the p-orbital but has one lobe significantly larger than the other.

The four hybrid orbitals of Carbon now spread away from each other to avoid any interelectronic repulsions. The angle at which the repulsions are minimum is 109.5o.
Bond Formation
The hybrid orbitals with one unpaired electron overlap with the electron present in the atomic orbital of the incoming atoms to form a single covalent bond or the sigma bond. The Carbon mainly forms such sigma bonds with the other elements of the p-block- Hydrogen, Oxygen, Halogens, Carbon, etc.
The bond formation reaction releases energy. The energy release compensates for the energy absorbed in the excitation process (s to the p-orbital). The stimulus is the formation of four new bonds for the Carbon having a tetrahedral shape with minimum electron repulsions.

As one s and three p orbitals were involved in the excitation, hybridization, and subsequent bond formation, such a Carbon is called sp3 hybridized carbon.
(Read the definition of sp3 hybridized carbon)
Examples of other sp3 hybridized molecules are- ethane (C2H6), water (H2O), ammonia (NH3), phosphorus trichloride (PCl3), etc.
These molecules may have different shapes and bond angles (other than tetrahedral and 109.5o) to avoid inter-electronic repulsions arising from the lone pair and the bond pair electrons.
