The bond angle is the angle at which two adjacent bonds converge and meet at the central atom in molecules.
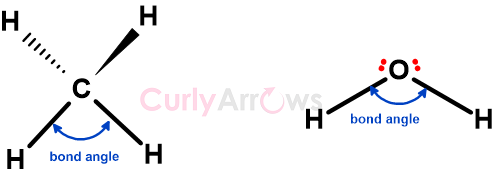
For example, the angle between H-C-H in the CH4 molecule is the bond angle, and C is the central atom. Similarly, the H-O-H is the bond angle in water, with Oxygen being the central atom.
The bond angles are measured in degree (denoted by the o sign in the superscript). So, the bond angle of CH4 is 109.5o, and that of H2O is 104.5o
How are bond angles formed?
VSEPR theory credits bond angle to the covalent bond formation process. According to the theory, when an atom (also known as the central atom) uses its valence electrons to form maximum covalent bonds with other atoms, it does so by undergoing a hybridization process.
The hybridization process is necessary since the valence electrons of the central atom may be present in different orbitals (like s, p, d) having varied energy levels (like 2s, 3p, 3d).
By the hybridization process, the atoms’ orbitals containing the valence electrons mix to form equal energy hybrid orbitals, so all the bonds that form out of these will have the same characteristics.
For example, carbon’s valence electrons are in different but closer energy levels, which mix to form four equal energy hybrid orbitals before forming covalent bonds with other atoms.
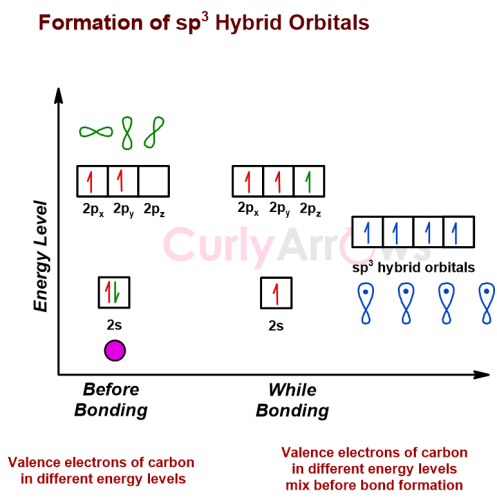
Any valence electrons that did not participate in the bonding process are left out as non-bonding electrons, also called the lone pair.
The bonding forming electron and lone pairs (if any) must then rearrange around the central atom to avoid crowding and interelectronic repulsions.
So, the central atom will adopt an arrangement that minimizes the electron pair repulsions by maximizing the distance between them, thereby creating bond angles.
Below is the tetrahedral shape that the carbon atom takes to form a CH4 molecule.
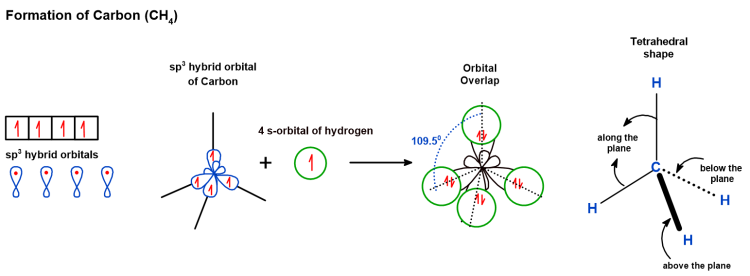
These hybrid orbitals then overlap with the orbitals of other atoms, the substituents, to form the maximum number of covalent bonds.
So, the hybridization of atoms is the most useful rationale for molecular geometry and their observed shapes.
In molecules, various bond angles and subsequent shapes are observed. When the bonds are fully spread, the repulsion is minimal at 180o, and the molecule adopts a liner shape. The repulsion is maximum when the bond angles are less than 90o. The bond angle of 72o is also seen in polyatomic molecules having considerable strain, for instance, IF7.
