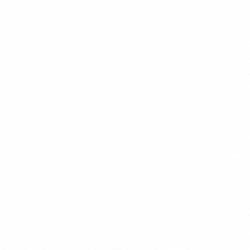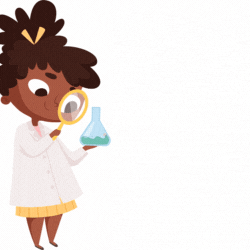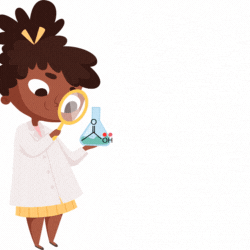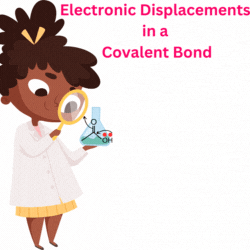The replacement of the hydrogen in acetic acid (H-CH2-COOH) with the chloro gives chloroacetic acid (Cl-CH2-COOH). Therefore, Acetic acid and Chloroacetic acid have carboxylic acid (R-COOH) as the principal functional group.
When a carboxylic acid (R-COOH) is placed in an aqueous medium, the molecule ionizes into an anion (RCOO-) and a proton (H+). The anion is called the conjugate base of the acid, RCOOH.
Ionization of carboxylic acid in an aqueous medium
R-CO-O-H (acid) → R-COO- (conjugate base) + H + (proton)
The carboxylic acid strength depends upon two factors-
a) ease of proton (H+) liberation
b) stabilization of the anion
Ease of proton liberation-
The hydrogen attached to heteroatoms (NH, OH, HX, etc.) is acidic, which means they are liberated as H+ ions. It is H+ that makes a solution acidic hence the name, acidic protons.
An acid can quickly liberate the hydrogens if the strength of the heteroatom-hydrogen bond is decreased. This is accomplished by introducing an electron-withdrawing group (such as Chloro) at the carbon next to the carboxylic acid functional group.
The attached chlorine atom is more electronegative than the carbon, due to which it engages in a negative Inductive effect (electron-withdrawing -I effect). The chloro pulls the electron density away from the oxygen, weakening the O-H bond and liberating the H+ ions quickly.

The acid strength is measured in pKa, where the lower values correspond to stronger acid.
Anion Stability-
The anion (-COO-) formed is resonance stabilized with the negative charge delocalized over the two oxygen atoms.
In addition to the delocalization over the oxygen atoms, the chloro spreads the negative charge over a larger distance due to its- I effect. The dispersal of negative charge over a large distance adds to the conjugate base stability of Cl-CH2-COO-.
In the case of the conjugate base of the acetic acid (CH3-COO-), the methyl group does not disperse the electron density. In contrast to the electron-withdrawing nature of chloro, methyl is electron-donating in nature (+ I effect). Methyl donates more electrons to add to the electron density that destabilizes the anion. Therefore, the CH3COOH prefers to exist mainly in the more stable unionized form.

Mechanism of proton loss and resonance stabilization-
The mechanism of the proton loss is the same for acetic acid and chloroacetic acid.
The divalent oxygen lone pair delocalizes over three atoms of the -COOH functional group by resonance. Due to the electron push, the hydroxy oxygen now carries a positive charge. The positive charge on an electronegative atom is highly unstable. Therefore, the oxygen pulls the electron density of the O-H bond and breaks it. The hydrogen is lost, and stable divalent oxygen is obtained, splitting the molecule into a cation (proton) and an anion (RCOO-).
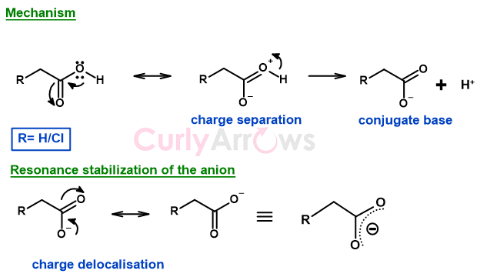
The anion is stabilized by resonance delocalization and -I effect in chloroacetic acid.
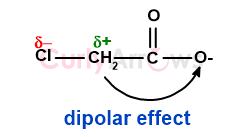
Chloroacetic acid has additional dipolar stability due to the polar C-Cl bond where the carbon has a partial positive charge and the chlorine a partial negative charge. The positive charge on the carbon causes it to attract the negatively charged anionic oxygen more strongly by the inductive effect, further adding to the stability. This intramolecular coulombic attraction known as the dipolar effect only exists for chloroacetic acid and not acetic acid.
Therefore, though the mechanism of the proton loss is the same for acetic acid and chloroacetic acid, it is the stability of the conjugate base that becomes the primary factor determining the acid strength.
The combination of -Ease of liberation of H+, stabilization of the negatively charged conjugate base over a larger area due to the -I effect of Chloro and the dipolar effect makes chloroacetic acid, a stronger acid over acetic acid.
The acidity of other chloroacetic acids
1) The dichloro and trichloroacetic acid will be a stronger acid than chloroacetic acid (increase in the number of electron-withdrawing groups)
2) Changing the position of chloro from the alpha (2-chlorobutanoic acid) to the beta (3-chlorobutanoic acid) or the gamma (4-chlorobutanoic acid) position will lower the acid strength.
Farther is the electron-withdrawing group/s, weaker is the inductive stabilization of the anion, and lower is the acid strength.
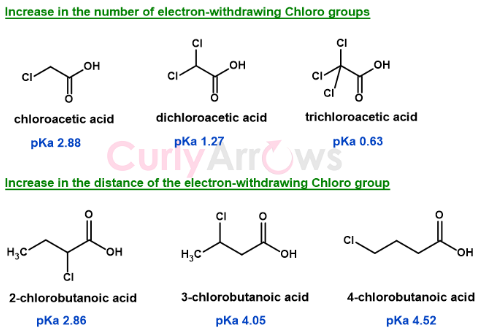
Note: The carbon next to a functional group is called the alpha carbon and the hydrogens alpha hydrogens. The carbon second and third to the functional group is the beta and the gamma carbons and hydrogens respectively.