Differentiation between Crystalline and Amorphous solids based on the Cleavage Property
If a crystalline solid is cut with a sharp object, it would always give parts with smooth edges whereas an amorphous solid would cut into surfaces with rough, uneven edges. Crystalline solids are therefore said to have cleavage property, and amorphous solids do not show cleavage property.
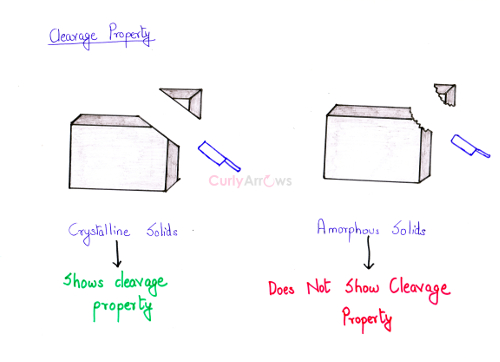
Imagine you are passing a raw potato through a slicer blade; what you get is thin slices of potato with smooth edges. But if you pass a boiled potato through a slicer, it would crumble and fall apart giving small uneven pieces with rough edges. The raw potato represents a crystalline solid and the boiled potato acts like an amorphous solid.
Why Crystalline Solids show Cleavage Property?
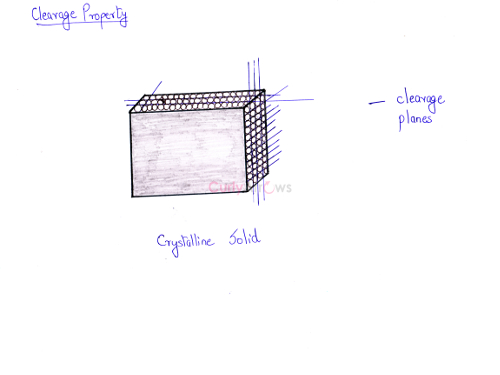
The cleavage property is shown by crystalline solids because they possess cleavage planes. In a crystalline solid, the cells are neatly stacked. The cleavage planes are areas where the crystal structure is the weakest. It is only along these planes that a crystalline solid can be cut. Therefore, a cut from a sharp object would give two smooth parts. Amorphous solids do not show any cleavage planes.
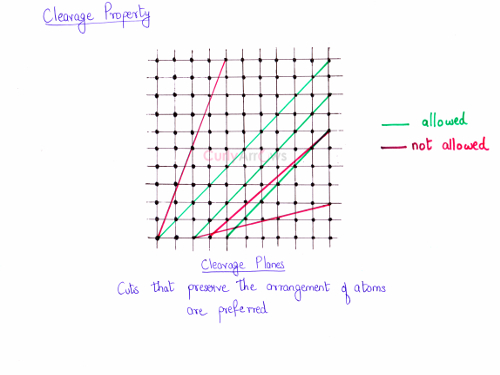
If you look at the crystal structure, you will notice a constant arrangement of the unit cells. In the two-dimensional diagram, it is seen as a lattice. Usually, the cuts made in the direction of the linear sequence of points shown in green are preferred over other cuts. The cuts along the direction of red are not allowed. These are the cuts that can shatter or disintegrate the crystal. In other words, the cuts that preserve the arrangement of the particles are preferred in a crystalline solid.
An example is of the diamond, a crystalline solid that when cut along the cleavage planes gives small diamonds with smooth edges having the same arrangement of the particles as that of the parent diamond.

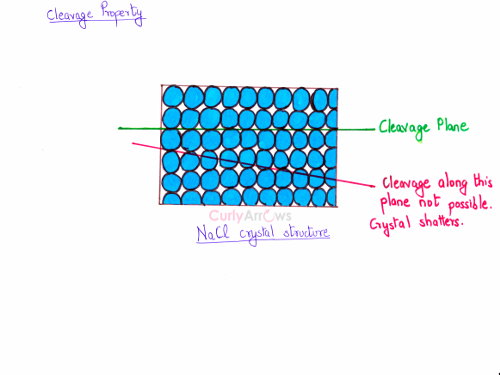
Let me give you another example. A NaCl crystal structure has a cubic arrangement. Its cleavage plane is parallel to the cube faces. If cut along any other plane as shown in red, the crystal structure will shatter.
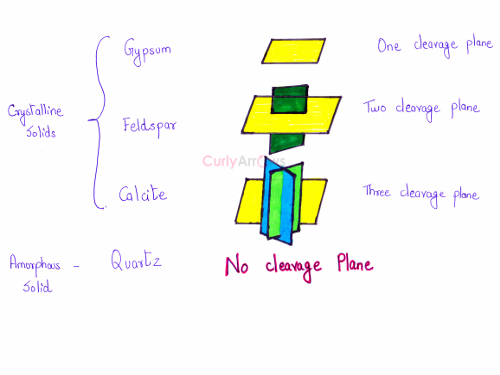
Every crystal has a unique cleavage plane depending on the arrangement of the particles. For example, crystals gypsum, feldspar and calcite have one, two and three cleavage planes.
The amorphous solid quartz does not have well-defined cleavage planes. It does not show cleavage property and breaks unevenly giving rough edges.
In summary, crystalline solids show cleavage property and is the reason why crystalline solids are smoother than the amorphous solids. Cut along the cleavage plane results in getting crystalline solid with smooth edges. Cleavage planes exist due to the ordered arrangement of the atoms thereby giving smaller crystalline solids of same geometric arrangement as the parent. On the other hand, the constituent particles of the amorphous solids are randomly arranged and do not show cleavage property. They break into uneven pieces with rough edges when cut.
.png)