On the dissolution of a substance in a dispersion medium, the aggregation of a vast number of atoms or smaller molecules of varying sizes (< 10-9 m) leads to the formation of colloidal particles of the size range 10-9 to 10-6 m.
As the colloidal particles are now made up of many atoms or molecules, it is now called multimolecular colloids.
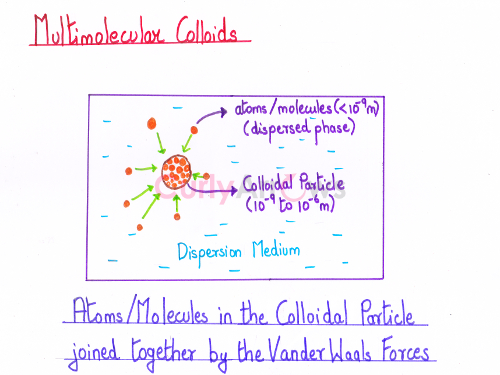
For example, a multimolecular colloid formed of a vast number of gold atoms of different sizes in a gold sol.
Sulfur sol made of hundreds of S8 molecules held together by Vander Waals forces.
.png)