Threshold energy is the minimum kinetic energy the molecules must have to bring about effective collisions between two reactant molecules considered as hard spheres, resulting in a chemical reaction.
Therefore,
Threshold energy= Average of the initial kinetic energy possessed by the reactants + Activation energy (Ea)
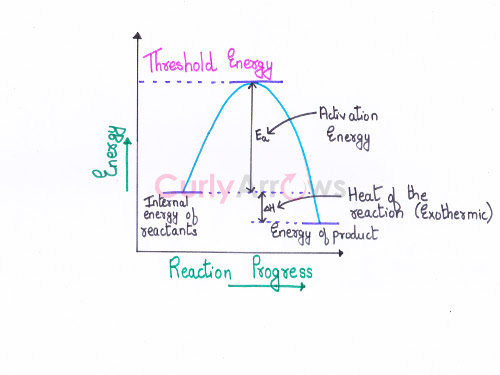
If the reactants at the start of the reaction have sufficient energy for the collisions, then the threshold energy is equal to the activation energy (Ea).
.png)