The electrochemical series or the activity series is the arrangement of various electrodes in the increasing order of their standard reduction potential. The reduction potential values are seen to increase from negative to zero and positive real numbers.
When arranged linearly, it would look like a number line with the positive numbers on the right side of zero and the negative numbers on the left side of zero.
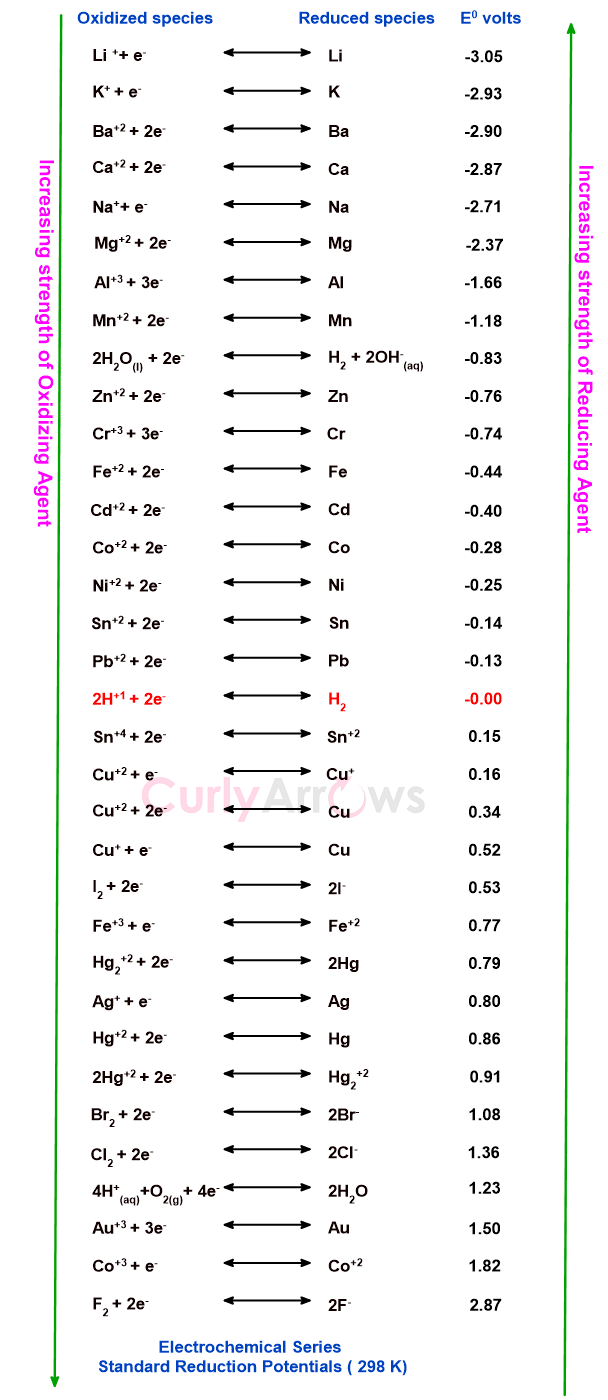
The reverse order will be seen if you arrange the electrodes as per their standard oxidation potential. It is because the electrochemical series are expressed as a reduction-oxidation equilibrium reaction.
The hydrogen electrode under the standard conditions of 1 M electrolyte concentration, temperature of 298 K and gas pressure of 1 atm is said to have zero reduction potential. The positive numbers are for the electrodes that undergo faster reduction with F2 having the highest reduction potential in the series. Similarly, the negative numbers are for the electrodes that do not undergo more rapid reduction (but undergoes rapid oxidation).
Li+ ion has the lowest reduction potential in the electrochemical series. Or we can say the reverse, that Li has stronger oxidation potential. Li+ ion will gain an electron or Li electrode will lose an electron.
Therefore, F2 with the highest reduction potential is a stronger oxidizing agent (gains the electrons to form 2F-) and Li with the lowest reduction potential is a more powerful reducing agent (loses an electron to give Li+ ions). Remember, an oxidizing agent is a substance that can bring about oxidation with itself undergoing reduction. It is an agent that will take the electrons by making a substance give up its electron and form the cation. Like a robber snatching your wallet. The wallet was yours until the robber took it and made it his own.
The electrochemical series can be memorized using a simple learning tool in the order of increasing strength of the reducing agent.
Mnemonic for Electrochemical series
Priyanka Chopra Sees Movie About Zebra In The Libya Hiring Cobra Studying Algebra.
Potassium > Calcium > Sodium > Magnesium > Aluminium > Zinc > Iron > Tin > Lead > Hydrogen > Copper > Silver > Gold (Au)
The most reactive are the group 1 and group 2 metals followed by the transition metal series that are least reactive towards oxidation. Hydrogen, a non-metal is taken as zero.
Read about the Importance and Applications of Electrochemical Series (With Examples)
.png)