An electrode when in contact with an electrolyte solution of the similar ionic nature (Example, Cu electrode in CuSO4 solution, Zn electrode in ZnSO4 solution) tends to either undergo Oxidation (loss of electrons) or reduction (gain of electrons).
Due to this oxidation or reduction, there develops a charge separation between the metal electrode and its ions in the solution creating a potential difference.

The potential difference is called the electrode potential. If the electrode undergoes oxidation, it is said to have oxidation potential and if it undergoes reduction, the electrode is said to have a reduction potential. The electrode potential when calculated under standard conditions of electrolyte concentration (1M), temperature (298K) and gas pressure (1 atm for gaseous reactions), it is called the standard electrode potential (Eo).
For an electrochemical cell, the electrode potential by convention is expressed as a reduction potential (Eored). As we know that the reduction half reaction and oxidation half reaction are just opposite of each other; the oxidation potential (Eoox) is therefore derived by reversing the sign of the obtained reduction potential and vice-versa.
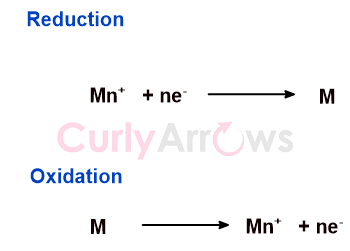
For Example
The oxidation electrode potential of a Zn electrode is expressed as EZn,Zn2+= 0.76 V. Its reduction electrode potential as per the convention should be written as,
EZn2+,Zn = - 0.76 V.
The standard reduction potential of a Hydrogen electrode is taken as zero, and it is used as a reference electrode to calculate reduction potential of any unknown electrode.
For Example
Consider two half-cells - one consisting of copper electrode dipped in 1M CuSO4 solution and a standard hydrogen electrode (SHE) as the other.
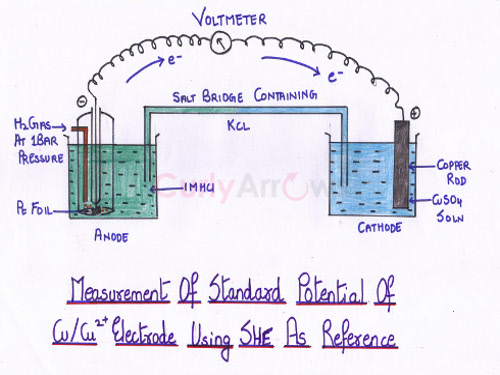
First, we must find out the electrode potential of the copper electrode; whether it is an oxidation or a reduction terminal. The direction of the flow of the current is the one that indicates whether at the copper electrode oxidation or reduction takes place. The voltmeter would give a reading only when terminals are attached correctly. The positive terminal of the voltmeter should be connected to the positive electrode, and the negative terminal should be attached to the negative electrode. Remember the mnemonic for ANOde- Anode is the Negative terminal where Oxidation takes place. On the right attachment, the voltmeter would give a reading and based on the terminal to which the copper electrode is attached; we can know if it is the cathode or the anode.
The EMF reading of the copper half-cell obtained is 0.34 V. The direction of the flow of current indicates copper terminal to be the cathode, a site for reduction reaction. Thus, 0.34 V is the standard reduction potential of Cu2+/Cu electrode.
The second example is a zinc electrode attached to SHE.
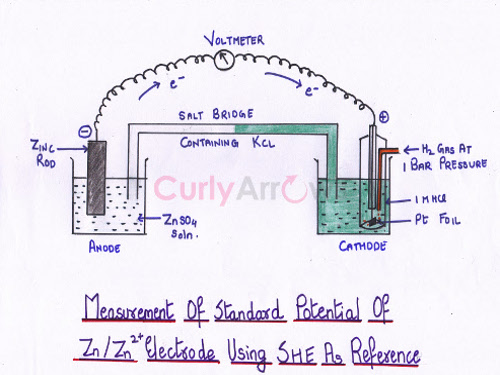
First, we must know whether it’s an oxidation terminal or reduction. The EMF of the cell obtained is 0.76 V. The direction of the flow of current indicates the zinc terminal to be the anode, a site for oxidation.
Since, Reduction potential = - Oxidation Potential
Thus, the standard reduction potential of the Zinc electrode obtained is - 0.76 V.
.png)