The Lewis dot structures are used to show the shared electron pairs between the bonded atoms in the molecule and the lone pairs of electrons if any. The representative structures follow the octet rule wherein the atoms combine by either transfer of electrons (loss or gain) or by sharing of valence electrons in a way that the valence shell of the atoms attains the octet configuration.
The electrons involved are shown as dots. A single bond is made up of two electrons, a double four and a triple bond six. Similarly, an atom can have one, two or three lone pairs of electrons.
The rules employed to draw a correct Lewis Structure are:
1) The least electronegative atom is the central atom.
Few Examples:
Molecule | Central Atom |
|---|---|
CO2 BCl3 SO42- | C B S |
2) The Hydrogen tries to attain the duplet and not the octet configuration. Therefore, it occupies the end or the terminal positions as shown in the images.
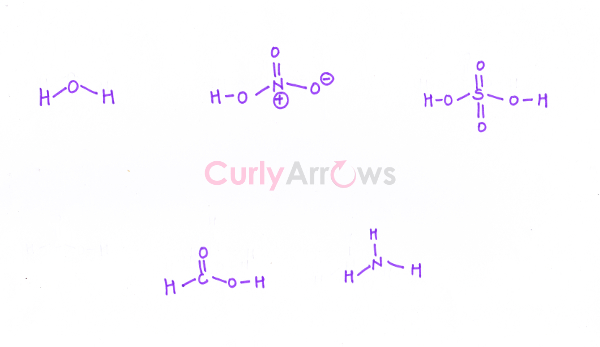
3) The valence electrons of all the atoms in the neutral molecule are added up. If the molecule is an anion, the charge carried by the anion is added up as electrons. Similarly, for the cations, depending on the charge carried by the molecule, the equivalent number of the electrons are deducted from the final answer.
Example,
Molecules | Valence Electrons | Addition of the valence electrons |
|---|---|---|
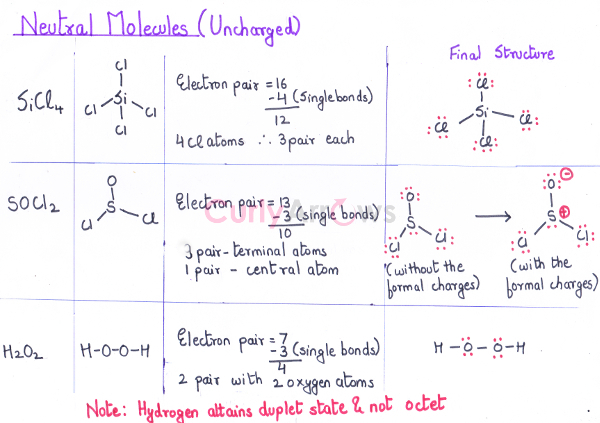
Neutral SiCl4 SOCl2 H2O2
|
Si -4, Cl- 7 S - 6, O - 6, Cl - 7 H - 1, O - 6 |
4 x1+ 7 x 4 (for 4 Cl atoms) = 32 6x1 + 6x1 + 2 x 7= 26 2 x 1 + 6 x 2 = 14 |
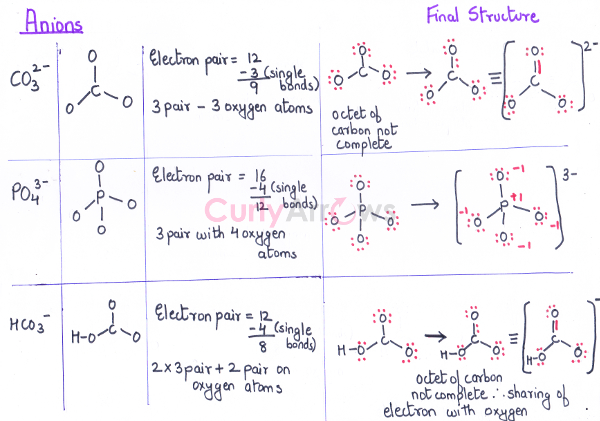
Anion CO32-
PO43-
HCO3-
|
C - 4, O – 6
P - 5, O – 6
H - 1, C - 4, O – 6 |
4 x 1+ 3 x 6 + 2 (for -2 charge) = 24
5 x 1 + 4 x 6 + 3 (for -3 charge)= 32
1 + 4 + 3 x 6 + 1 (for -1 charge) = 24 |
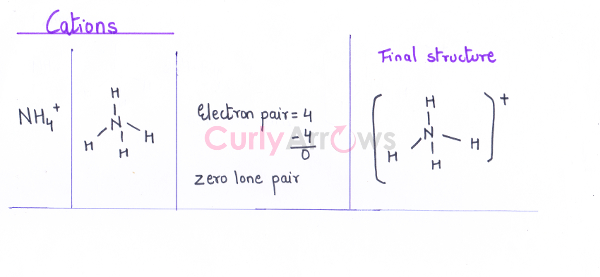
Cations NH4+
|
N – 5, H - 1 |
5 x 1 + 4 x 1 – 1 (for +1 charge)= 8 |
4) Divide the number of electrons obtained from step 3 by 2 to get the electron pairs. It is easier to assign Lewis dots when the electrons are paired. Note that odd electrons disobey the octet rule.
For Example,
Molecules | Valence Electrons | Addition of the valence electrons | Valence Electrons Divided by 2 = Electron Pairs |
|---|---|---|---|
Neutral SiCl4
SOCl2
H2O2
|
Si -4, Cl- 7
S- 6, O - 6, Cl - 7
H - 1, O - 6 |
4+ 7 x 4 (4 Cl atoms) = 32
6 + 6 + 2 x 7= 26
2 x 1 + 6 x 2 = 14 |
32/2 = 16
26/2 = 13
14/2= 7 |
Anion CO32-
PO43-
HCO3-
|
C - 4, O – 6
P - 5, O – 6
H - 1, C - 4, O – 6 |
4+ 3 x 6 + 2 (for 2 electrons) = 24
5 + 4 x 6 + 3 = 32
1 + 4 + 3 x 6 + 1 = 24 |
24/2= 12
32/2= 16
24/2= 12 |
Cations NH4+
|
N – 5, H - 1 |
5 + 4 x 1 – 1 = 8 |
8/2 = 4 |
5) Keeping the least electronegative atom as the central atom, attach the atoms with each other using a single bond. One bond is one electron pair. Deduct the number of single bonds from the electron pairs.
The remaining unshared pair of the electrons are used as lone pairs (first satisfying the terminal atoms and then the central atom in a way that every atom has an octet configuration) or in multiple bonding.
Example, Neutral Molecules (Uncharged)
Example, Anions
Example, Cations