In the presence of moisture, PCl3 undergoes hydrolysis to release fumes of HCl gas.
PCl3 + 3H2O → H3PO3 + 3HCl
The reaction mechanism is shown as,
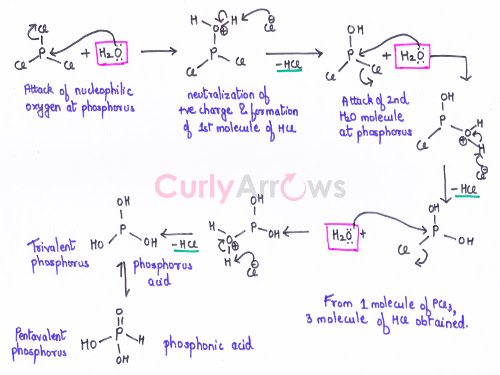
In the first Step, the nucleophilic Oxygen of the water molecule (present in moisture) attacks the Phosphorous followed by the loss of Cl- ion.
In the next step, the Cl- ion abstracts the proton and neutralizes the positive charge on the Oxygen thereby, generating the first molecule of HCl. The process is repeated two more times for the other two Chlorine atoms of PCl3 molecule.
In the end, the trivalent Phosphorous undergoes rearrangement to the more dominant pentavalent tautomeric form, generating Phosphonic Acid (H3PO3).
.png)