In Lewis Structures, the valence electrons are shown as dots. When atoms combine to form a bond, it is the electrons that participate. For Ionic Bonding there is a transfer of electrons (loss or gain), and for covalent bonding, the electrons are shared between the atoms. The atoms do so to attain the stable octet configuration of 8 electrons in their valence shell.
The Lewis diagrams show the bonded electrons and the lone pair of electrons as dots. The electronic distribution of the entire molecule can be understood by looking at the Lewis Structures. If the octet of the bonded atoms is incomplete, the atoms then share the remaining electrons to form double or triple bonds. The unused electrons are then shown as lone pairs.
Lewis structures can also be known as Lewis dot diagrams, Lewis dot formulas, Lewis dot structures, and Electron dot structures.
Example,
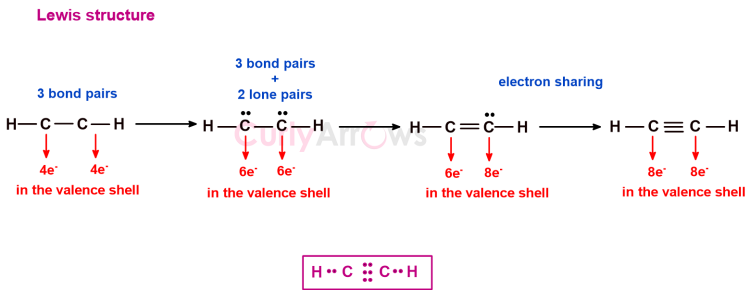
Draw Lewis Structure of C2H2
Steps to draw Lewis Structure:
1) Calculate the Valence Electrons
Valence Electron of C-4, H-1
C2H2= 2 x 4 + 2 x 1= 10
2) Obtain the electron pairs
Valence Electrons/ 2= 10/2 = 5
3) Assign the single bonds
H-C-C-H
4) Deduct the single bonds from the Electron pairs
Three Single bonds, therefore 5-3= 2 electron pairs remaining
5) Assign the Lone pairs
The two remaining electrons will be shown as the lone pairs
6) Share of the Electrons to obtain the octet