For an electrode in contact with an electrolyte solution having similar ionic nature, if the tendency of the electrode is to lose electrons and undergo oxidation reaction, the electrode is said to have an oxidation potential. Example, Zn electrode dipped in the ZnSO4 electrolyte solution.
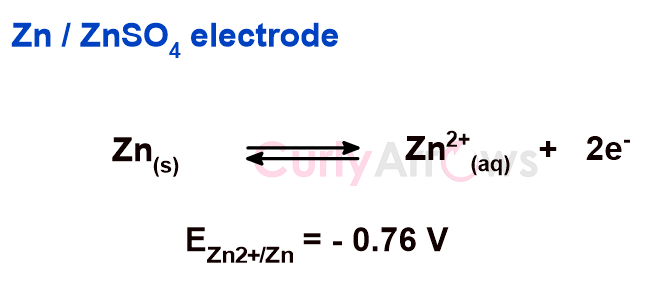
If the oxidation potential is calculated under the standard condition of 1M electrolyte concentration, 298 K temperature, and 1 bar atmospheric pressure (if gas is involved), it is called standard oxidation potential. The standard oxidation potential is represented as Eoox.
The standard oxidation potential can be derived using standard hydrogen electrode (SHE) as the reference electrode.
.png)