A pentahalide or a trihalide represents an element of group 15 with five or three halogen atoms.
An element must either share electrons by covalent bonding with the halogens or lose electrons and ionic bond with the halogens to form a pentahalide or a trihalide.
The elements in Group 15 of the periodic table are- Nitrogen (N), Phosphorus (P), Arsenic (As), Antimony (Sb), and Bismuth (Bi).
(Remember the mnemonic- New Popes Assign Subordinate Bishops)
The elements of Group 15 have a general electronic configuration of ns2 np3 where n=2 to 6.
The group 15 elements show two common oxidation states- +5 and +3 by losing outermost valence electrons from the s and p orbitals.
Neutral element: [core electrons] ns2 np3
+3 oxidation state element: [core electrons] ns2 np0
+5 oxidation state element: [core electrons] ns0 np0
Therefore, the Group 15 elements should be able to form ionic compounds by losing 3 and 5 outer electrons. However, it is observed that they mostly form covalent and not ionic compounds.
In the periodic table from N to Bi, the atomic size of the element increases. As the atomic size increases, more orbitals are involved in accommodating the increase in the number of electrons. From phosphorus onwards, the d and f orbitals increase an atom’s size.
These d and f orbitals are diffused with the electrons further spread out than in s and p orbitals. Therefore, the inner d and f-orbitals do not effectively shield the outermost orbital electrons (ns and np) from the attractive nuclear pull. Due to the lack of an umbrella kind of protection, the nucleus tightly holds the s-orbitals and does not lose electrons to achieve a +5-oxidation state. Therefore, the s-orbital electrons remain as an inert pair in ionization reactions.
Therefore, due to ns2 electrons’ inert pair effect, the elements that have d and f-orbital involvement can only lose the p-electrons. The elements show only a +3-oxidation state to form M3+ ions. For example, BiF3 and SbF3exist as ionic solids.
The electronic configuration of Sb and Bi are-
Sb: [Kr] 4d10 5s2 5p3 Bi: [Xe] 4f14 5d10 6s2 6p3
Sb+3: [Kr] 4d10 5s2 5p0 Bi+3: [Xe] 4f14 5d10 6s2 6p0
The +5 oxidation state halides are unknown for N. Stability of +5 oxidation state decreases from P to Bi. Bi shows +5 oxidation state with only high electronegative Fluorine to form BiF5.
Therefore, when s electrons remain paired, the oxidation state is always two lower than the usual oxidation state of the group.
Most compounds formed by the group are covalent since it requires too much energy to lose the outer three and five electrons to form M+5 and M3+ ions.
Nitrogen forms three-sigma σ bonds using its unpaired electrons with three other atoms forming a tetrahedral shape. The lone pair of electrons occupy one position in the tetrahedron.
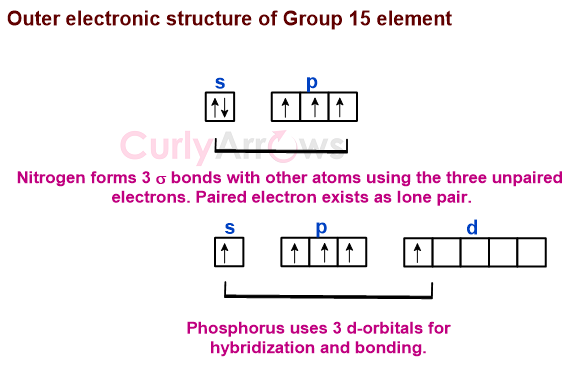
Phosphorus forms pentavalent compounds with other elements by involving one or two d-orbitals in bonding —for example, PCl5 and PF5.
Other covalent compounds are NCl3, PCl3, AsCl3, AsCl5, and SbCl3.
In conclusion, the Group 15 elements prefer a +3-oxidation state to form ionic compounds or trihalides due to the inert pair effect. The +5-oxidation state is rare for some elements and only forms with highly electronegative atoms like Fluorine. The group 15 elements, therefore, prefer hybridization to form covalent pentahalides compounds.
.png)