The Hydrogens attached to a Carbon atom are said to be equivalent if they are in a same chemical environment. Same chemical environment means that under a reaction condition, these hydrogens would lose their identities of being attached to separate Carbons and behave like an identical set.
Identifying such hydrogen types would help in predicting the number of monohalogen products that could be obtained on free radical halogenation reaction.
For example, in neo-pentane, there are twelve hydrogens in a set of four attached to four Carbons.
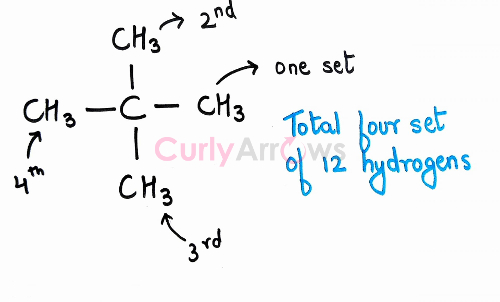
All the Hydrogens in a set are identical, but the aim is to find whether the hydrogens in different sets are also similar to give equivalent sets. For finding equivalent sets of Hydrogen, a hypothetical plane called a plane of symmetry is passed along the x, y, and the z-axis that divides the molecule into two equal parts. If the divided parts look like mirror images of each other and show symmetry, they are called an equivalent set.
In the examples shown below, for simplicity, we will use a pencil for an imaginary passing plane.
In neo-pentane, a plane is passed through the Carbon atoms numbered 2, 4 and 5 along the y-axis, we see that the two methyls (C1 and C3) are structurally similar and that would give us one set of chemically equivalent hydrogens.
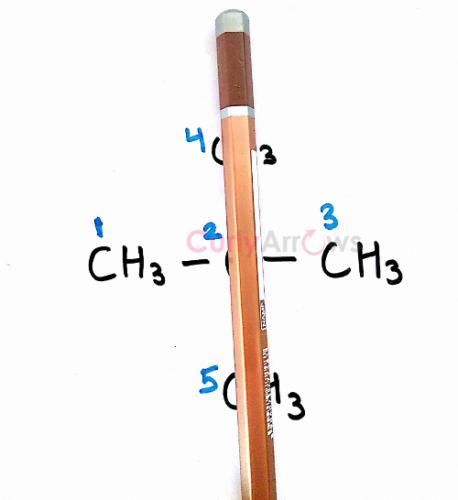
Similarly, on passing the pencil through the Carbon atoms numbered 1, 2 and 3 along the x-axis, we obtain another set of equivalent Hydrogens.
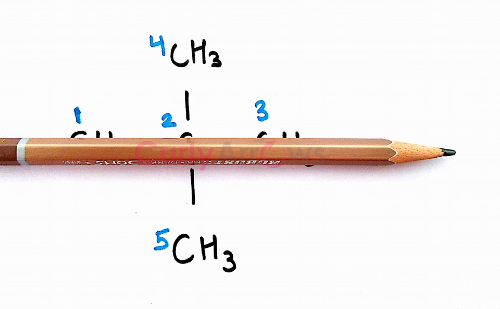
So, the
Hydrogens of Carbon (C1) = Hydrogens of Carbon (C3) and
Hydrogens of Carbon (C4) = Hydrogens of Carbon (C5)
To prove, equivalency in the remaining Hydrogen sets of Carbon (C3) and Carbon (C4), Carbon (C1) and Carbon (C5), we will pass the plane between C3-C4 and C1-C5.
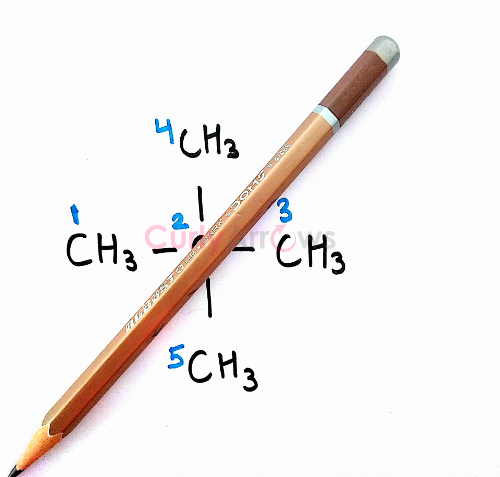
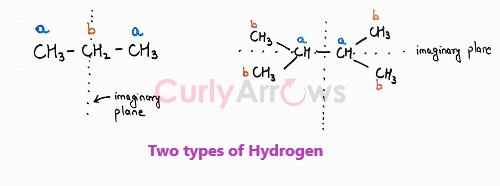
We see that all the methyls and the hydrogens are structurally equivalent. The four set of methyls will behave as a single set and on free radical halogenation reaction, only one monohalogen product would be obtained proving they are chemically equivalent as well.
Example 2: Isopentane
On passing the plane along the x-axis, we see that hydrogens of C4 and C5 are identical. But a plane that divides the molecule along the y-axis between each carbon atoms, a dissimilar set of non-equivalent Hydrogens are obtained. Therefore, the
Hydrogens of C1 ≠ C2 ≠ C3 ≠ C4/C5
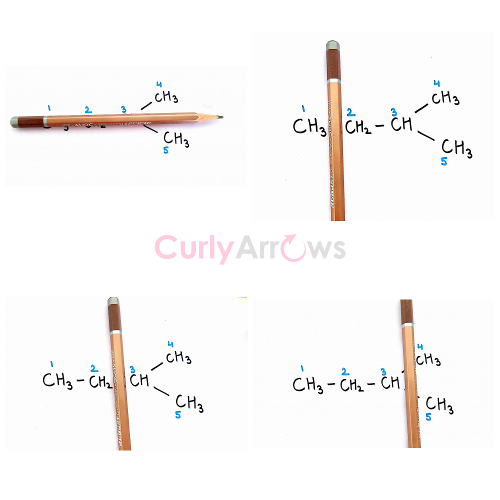

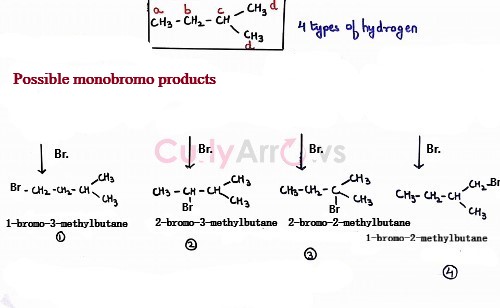
For isopentane, the types of Hydrogen are four -1CH3-, -2CH2-, -3CH- and -4,5CH3-. Such a molecule on free radical halogenation reaction would give four monohalogen products.
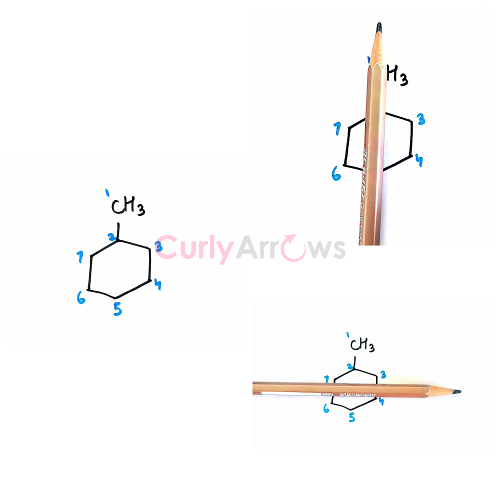
Example 3: 1-methylcyclohexane
Identical sets of Hydrogens are seen on passing the plane along C1, C2, and C5.
Hydrogens of Carbon 3 = Hydrogens of Carbon 7 and Hydrogens of Carbon 4 = Hydrogens of Carbon 6
Any other axes, we get dissimilar sets of Hydrogens. Therefore, for 1-methylcyclohexane, total sets of equivalent Hydrogen are five -1CH3-, -2CH-, -3,7CH2-, -4,6CH2- and -5CH2- .

Few more examples,
If the Hydrogen is attached to a heteroatom (-OH, -NH2, etc.) it would be treated as a separate set. At times to identify the equivalent hydrogens in addition to the plane of symmetry, the axis of rotation is also used wherein the molecule is rotated around an axis for finding symmetry.
For example, in case of tert-butyl chloride, if the axis of rotation is along the Chloro, then the three methyls are equivalent. The number of the equivalent set of hydrogen is one.
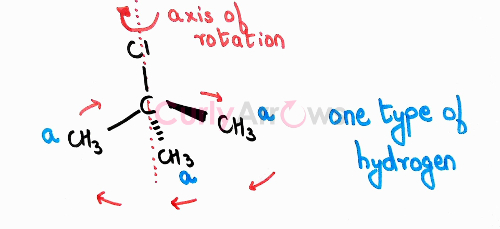
In summary, identifying types of hydrogen helps to predict the number of possible products that can be obtained on free radical halogenation reaction. Though, the major product formed would depend on the stability of the free radical. Identifying equivalent hydrogens also finds application in Nuclear Magnetic Resonance (NMR) studies. Here the importance is given to structurally equivalent Hydrogen (and not Carbon) as it is the Hydrogens that give NMR signals. For a 13C NMR, the focus would shift entirely on finding equivalent Carbons.
Practice Problem
Identify the types of equivalent and non-equivalent hydrogen (quiz)
.png)