While working on a fine morning in 1827, little did a botanist named Robert Brown know that what he was about to witness through his microscope would eventually lead to the discovery of the existence of invisible matter.
He witnessed bumping between particles when he carefully looked into his microscope to observe a slide of pollen grain in water. The motion was constant but zig-zag as though the pollens were striking into some invisible object and turning its course. This type of motion came to be known as the Brownian motion.
Later Einstein, in the year 1905, used the Brownian Motion for his Ph.D. thesis to prove the existence of smaller entities- atoms and molecules. He explained that the Brownian motion was the visible manifestation of the random movement of the microscopic pollen particles that strike the invisible molecules of water. The theory was later tested on other inorganic matter like soot, and they too followed the zig-zag motion.
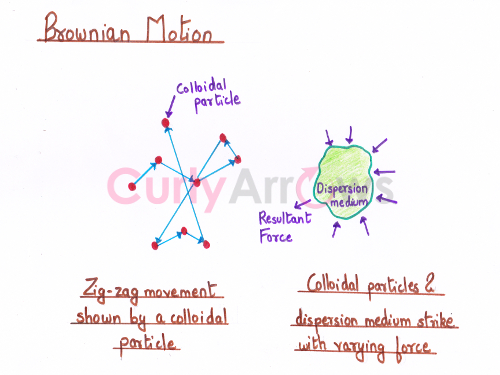
However, for a substance to show continuous zig-zag Brownian motion, the particle's size must be between 10-9 to 10-6 m while suspended in a dispersion medium. Such particles with that size range are called colloids, and the dispersion medium is called the colloidal sol.
The striking of the colloidal particles with the smaller molecules of the dispersion medium occurs with varying velocity and direction. This causes the colloidal particles to bounce off with different forces from the molecule's surface that drive such zig-zag motion.
Therefore, the particles move continuously and do not settle, creating a stirring type of effect. The stirring effect is responsible for the stability of the sol so that they do not separate into two parts- the solid particle and the liquid.
The Brownian motion is independent of the colloidal particle's nature but highly depends on the size of the particle. The bigger the size of the particle and higher the dispersion medium's viscosity, the slower the particle's motion.
The Brownian motion, therefore, explains the dynamic nature of the molecules present in a dispersion medium.
Importance of Brownian Motion
The study of the Brownian motion was instrumental in determining the Avogadro's Number (NA) and is also extended to the other branches of science -biology, physics, mathematics, evolution, and finance (stock market analysis).
.png)