If all the elements were to create a gated community for themselves; they would build four apartment blocks namely, the s-block, p-block, d-block and the f-block. The elements having similar property would be grouped together occupying one block and this behavior is based on the orbital the last electron chooses to enter.
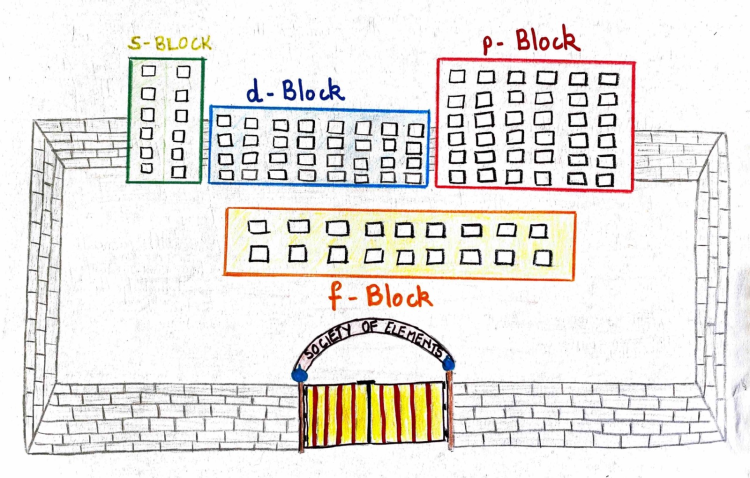
If the last electron enters the s-orbital, it belongs to the s-block and so is the case with the others. Hydrogen, Helium and few elements of the d and f-block are the exception to this rule.
The s-orbital can only accommodate two oppositely spinning electrons. For the Group I elements on the left side of the s-block; a single, unpaired electron enters the outer s-orbital giving a general electronic configuration of ns1 for all the elements in this group.
For the Group II elements, the second electron having opposite spin to the first enters the outermost s-orbital thus filling the orbital and giving a general electronic configuration of ns2.
Including the Hydrogen for Group I and Helium for Group II, there are 14 elements in total, seven on the either side forming part of the s-block.
The correct placement of Hydrogen (H) and Helium (He) in the periodic table is still a topic of debate. Hydrogen having an electronic configuration of 1s1 can be considered as a Group I s-block element and is placed alongside the alkali metals having the half filled general electronic configuration of ns1. It can also behave like the halogens of Group VII by accepting electrons to complete its valence shell configuration thereby attaining stable duplet state. Hydrogen is sometimes placed above Carbon in Group IV in addition to Group I and the Group VII of the periodic table.
Helium (He) can be considered as a Group II s-block occupant as its last electron enters the s-orbital but it can also be placed with the p-block elements in Group VIII having a complete shell and acting as an inert gas. Therefore, based on the electronic configuration, Helium (He) is a Group II s-block element but based on the property/nature, it is a Group VII p-block element placed with the inert gases.
Due to these reasons, at times, you might find the position of Hydrogen and Helium to be repeated twice in the periodic table or they may be placed separately at the centre of the periodic table.
Group I elements excluding the Hydrogen are called the alkali metals and Group II elements excluding the Helium are known as the alkaline earth metals.
The Alkali Metals
The alkali metals are Lithium (Li), Sodium (Na), Potassium (K), Rubidium (Rb), Caesium (Cs) and Francium (Fr). The mnemonic to remember all the alkali metals of the group I is:
Little Naughty Kids Rub Cat’s Fur
The alkali metals derived their name based on their reaction with water forming hydroxides that are strong bases or alkalies.
As Naughty Kids are found in abundance on the earth so are the elements Sodium (Na) and Potassium (K) followed by Hydrogen (H), Rubidium (Rb), Lithium (Li) and Caesium (Cs). Francium (Fr) is a radioactive element having a half-life of only 21 minutes.
The Alkaline Earth Metals
The alkaline earth metals are Beryllium (Be), Magnesium (Mg), Calcium (Ca), Strontium (Sr), Barium (Ba) and Radium (Ra). The mnemonic to remember all the elements of the group II:
Beer Mugs Can Serve Bar Rats
Unlike the alkali metals, the Alkaline Earth Metals does not exist in the free elemental state but are found as compounds on the surface of the earth thereby called the alkaline earth metals. They usually occur in the oxide form namely- beryllia (BeO), magnesia (MgO), lime (CaO), strontia (SrO), and baryta (BaO). These oxides on reaction with water generate hydroxides or alkalies, except Beryllium that remains unreactive.
Concerning the elemental abundance, Calcium (Ca) and Magnesium (Mg) are the 5th and the 8th most abundant element in the earth’s crust followed by Strontium (Sr) and Barium (Ba). Beryllium (Be) and Radium (Ra) are among the rare elements found in the igneous rocks of the earth’s crust.
.png)