- The unit cells that makes up the entire crystalline solid has a fixed value for the length of the edges and the angle between the edges.
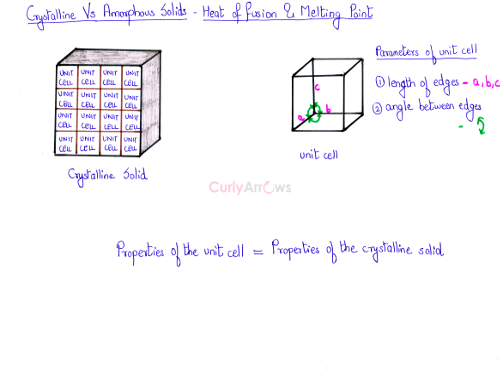
- Therefore, the heat energy required to melt a crystalline solid would have a fixed value.
- The energy required to bring about the change of state from a unit mass of the solid to the liquid is called the heat of fusion, and the temperature at which it happens is called the melting point.
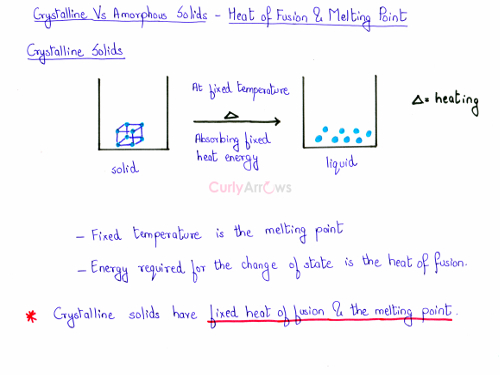
Read more about the Heat of Fusion and Melting Point- Crystalline versus Amorphous Solids
.png)