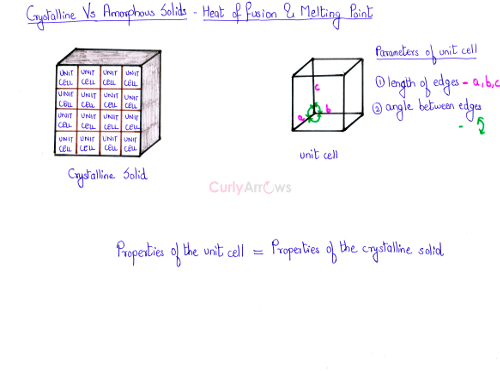
- As the Crystalline solids are made up of a large number of unit cells extending in all the three dimensions, properties of the unit cell dictate the properties of the crystalline solid.
- Each unit cell has a fixed value for the length of the edges and the angle between the edges.
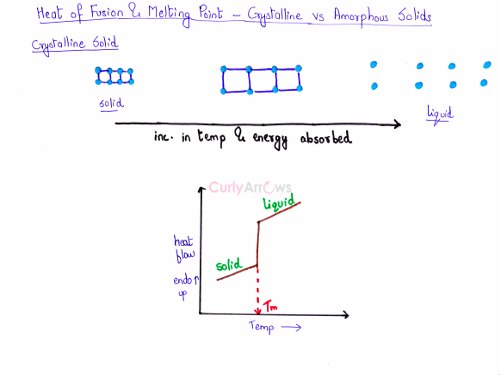
- On supplying heat energy, the unit cells would collapse at the same time at a fixed temperature, with a sharp transition from the solid to the liquid state. This temperature is the melting point of the crystalline solid (shown here as Tm).
Read more about the Heat of Fusion and Melting Point- Crystalline versus Amorphous Solids
.png)