A halogen derivative of an alkane is called a Haloalkane, where the hydrogen is replaced with a halogen.

The common or the trivial name is Alkyl Halide.
A fun mnemonic to remember a few common preparation methods of haloalkane is hidden in the common name.
Mnemonic : AAAlkyl Halide (Alkane, Alkene, Alcohol, Halogen transfer)
The preparation of a haloalkane (or the alkyl halide) can be done from starting materials alkane, alkene, alcohol, and replacement of halogens by halogen transfer reactions.
Alkanes are very stable, inert molecules that undergo only a few reactions - cracking (breaking down a larger carbon chain into smaller carbon fragments), combustion (oxidation of an alkane to give carbon dioxide and water molecules), and free radical halogenation reactions (replacing Hydrogen of an alkane with halogen).
Here the preparation of haloalkane from alkane using free radical halogenation method will be covered.
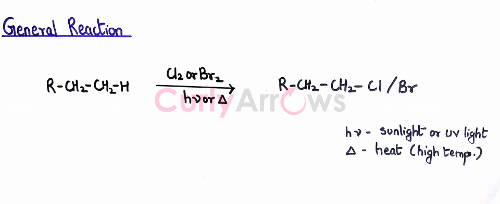
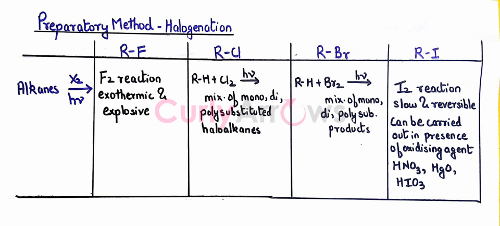
Preparation of Haloalkane from Alkanes- Free Radical Halogenation
General Reaction
For free radical halogenation, an alkane is reacted with the halogen in the presence of a catalyst, UV light, or sunlight. In the absence of a light source, a very high-temperature condition of 520 K -670 K is used.
Halogen Reactivity in Halogenation Reaction
The order of the halogen reactivity is-
F2 > Cl2 > Br2 > I2
Most halogenation reactions are done using Cl2 or Br2.
Reactions with F2 are highly reactive, explosive, and hard to control.
In contrast, the reactions with I2 are very sluggish and reversible. Reversibility is due to the formation of by-product HI along with the desired iodoalkane.
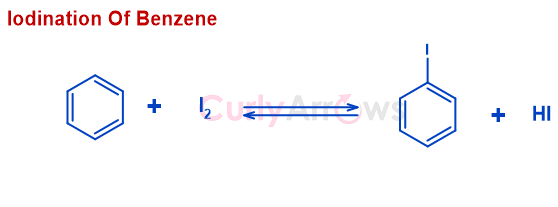
One solution is to use an oxidizing agent such as - HNO3, HgO, etc., converts HI formed back to I2. An increase in the I2 concentration pushes the reaction in the forward direction (according to the Le Chatelier principle) to obtain more iodoalkane. (Read why iodination of benzene is difficult)
Formation of polyhalogenated products- A serious concern
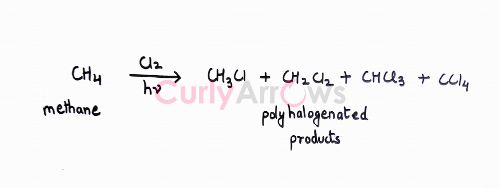
The halogenation reaction using Cl2 or Br2 gives a mixture of mono, di, and polyhalogenated products.
The number of products formed depends on the number and type of replaceable hydrogens.
For example, methane (CH4) has four hydrogens, and it can form four different (mono, di, tri, tetra) halogenated compounds.
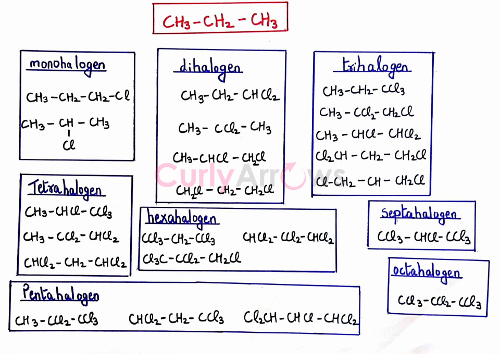
The maximum number of replaceable hydrogens for a slightly bigger carbon chain propane is eight. Therefore, there are twenty-two combinations of halogenated products that can be obtained in presence of excess halogen.
The formation of polyhalogenated compounds can be avoided if the alkane ratio to the halogen is decreased (1: 0.8 ratio) so that only a few halogens are available per molecule of alkane. The primary product would then be a mono halogen compound.
For a molecule like propane with more than two types of hydrogens (a and b), the issue doesn’t just stop there. There again is a possibility of forming two types of monohalogenated products.
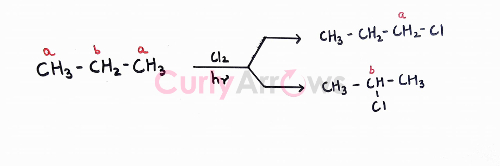
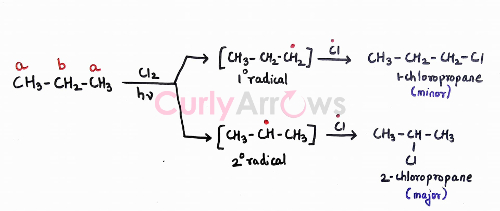
Remember that halogenation reaction with alkane goes via a free radical mechanism. A radical, an electron-deficient species, follows the stability order like the carbocations- 3o> 2o> 1o. Therefore, the major product will be the one where the radical formed before the attachment of the halogen is most stable.
For propane, 2o radical formed is more stable than 10 radical to giving 2-chloropropane as the major product. 1-chloropropane obtained from a less stable primary radical will be the minor product.
Also, note that 2-chloropropane and 1-chloropropane are positional isomers with close boiling points and would be hard to isolate.
Another example to explain the correlation between the type of the hydrogen and most stable product formed is for isopentane -
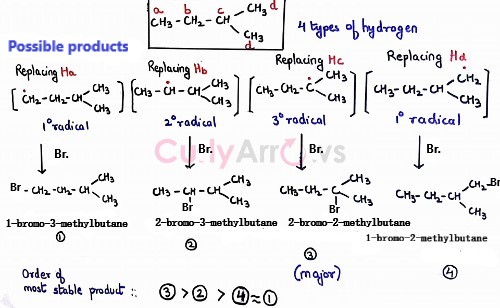
Isopentane has four types of hydrogen. However, 30 radical is the most stable, and the product formed using that will be the major product.
Therefore, selective halogenation for an alkane without giving side products is a major hurdle in free radical reactions.
Selectivity is seen for allylic or benzylic compounds where monohalogenation alkane is the major product.
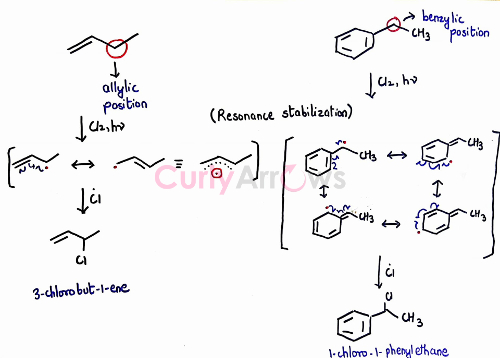
In summary, free-radical halogenation reactions are hard to control. A mixture of desired monosubstituted and polysubstituted products is formed, making the purification and isolation of the desired compound a tedious process. Therefore, free radical halogenation of alkane is not a preferred way to halogenate a compound.
For making a fluoro or an iodoalkane, halogen transfer reactions are preferred.
Related Reading: Free radical - Introduction, Structure, and Types, Types of equivalent hydrogen, Identification of allylic and benzylic position, Identification of 1o, 2o and 3o alkyl groups
Practice Problems
b) Identify the number of dihalogen derivatives of the following compounds (quiz)
c) Predict the major product of free-radical bromination reaction (quiz)
