Understanding the failure of Benzene to undergo an Iodination reaction requires insight into multiple aspects of the reaction mechanism and the nature of the reactants. Here in the article, we will explore a few methods of increasing the reactivity of Iodine using different reaction conditions.
Electrophilic Aromatic Substitution Reaction
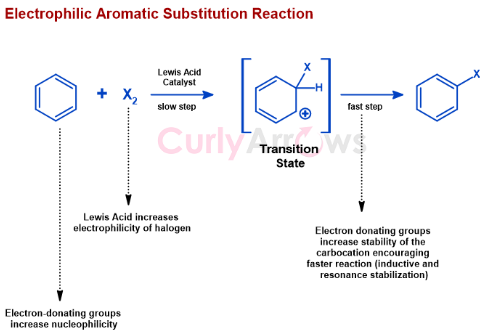
An electrophilic aromatic substitution involves an aromatic compound, usually Benzene, as the starting material. The hydrogen is displaced with an electrophile to form the final desired substituted Benzene product in the reaction.
For a faster electrophilic aromatic substitution reaction, any reactants, the Benzene, or the halogen, must be slightly more reactive. Reactivity of the Benzene is increased by making it more nucleophilic by introducing electron-donating groups. For the halogens, the Lewis acid catalyst increases its electrophilicity. These changes help achieve a faster transition state, which helps improve the reaction rate and form the product.
Role of the Halogens
Halogens (F, Cl, Br, I) naturally exist as diatomic molecules (F2, Cl2, Br2, and I2). These diatomic molecules are electrophilic; they are willing to accept electrons to form more stable anions.
X2 + 2e- → 2X-
Amongst the halogens, Fluorine is the most electronegative. It is more willing than others to accept the electrons and therefore is most electrophilic, whereas Iodine is the least. Thus, an electrophilic substitution reaction of Benzene with Fluorine is highly reactive, and Iodine is unreactive.
The reactivity can be correlated with the exothermic rate of the halogenation reaction. Fluorination reaction proceeds exothermically and explosively. The reaction cannot be controlled, resulting in polyfluorinated products. Whereas for Iodination, the reaction is endothermic (12kJ/mol of energy absorbed) and largely unreactive.

Iodination of Benzene
Under conventional reaction conditions, I2 adds to the Benzene reversibly generating HI. HI, is a strong reducing agent, and it regenerates I2 on reaction with the aryl iodide giving back the starting material, Benzene.

One way to obtain iodobenzene is to remove HI as soon as it is formed in the reaction mixture by forming salts. For example, when the Mercuric oxide is used, it converts Hydrogen Iodide to Mercuric Iodide, which is then discarded.
Other methods include using oxidizing agents. The oxidizing agents such as HIO3 convert HI to give Iodine, thereby increasing its concentration in the reaction mixture. This is in accordance with Le-Chatelier’s principle. It states that if the concentration of one of the reactants is increased, the equilibrium shifts in the forward direction to help increase the concentration of the product, aryl iodide.
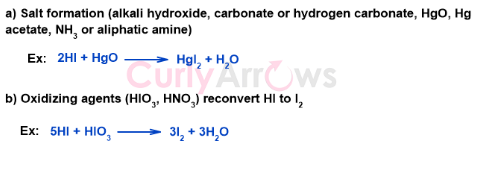
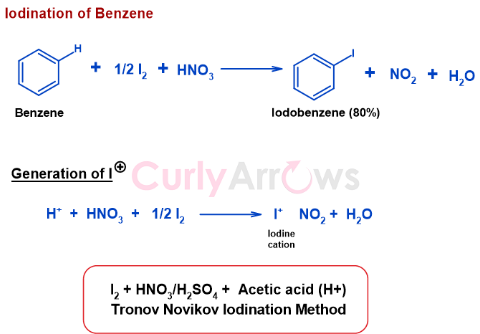
Oxidizing agents oxidize I2 to iodonium ion I+. The earliest reported reaction was the Tronov-Novikov reaction. It used HNO3 or H2SO4 in the presence of Acetic acid and Iodine to give iodonium ion. The cation then reacts with Benzene to give Iodobenzene, Nitrogen dioxide, and water.
A similar mechanism is seen for Cupric Chloride and Hydrogen peroxide for the generation of Iodine cation.

Other oxidizing agents are Oleum (H2SO4.SO3), Silver sulfate (Ag2SO4), Sodium periodate (NaIO4), Periodic acid (HIO4), Sodium hypochlorite (NaOCl), Iodic acid (HIO3), Nitric acid (HNO3) and Sulfuric acid (H2SO4), Sulfur trioxide (SO3), and Hydrogen peroxide (H2O2). Sometimes Lewis acid catalyst is used along with the oxidizing agents.
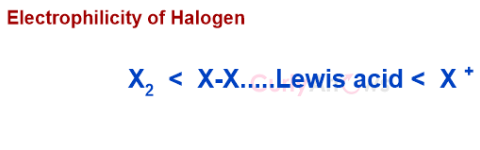
The formation of iodonium cation is more effective than the Lewis acid-halogen combination to convert non-reactive, non-polar Iodine into reactive species.
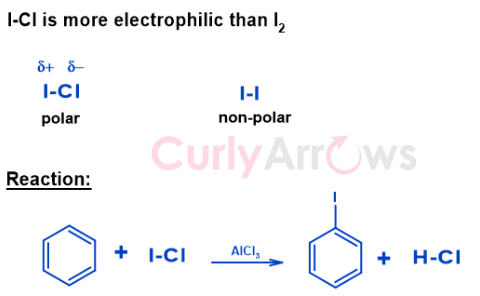
One of the reagents, Iodine monochloride ICl, an interhalogen compound, is effective for Iodination. Chlorine is more electronegative than Iodine and pulls the bond electrons towards itself. This imparts Iodine with a (delta) positive charge making it electrophilic. The Benzene ring can pick up this more quickly than Iodine from the non-polar I2 bond, giving aryl iodide the desired product.