Question- How many protons and electrons do the following species have?
Na+ (atomic number Z=11), Ar (atomic number Z=18), Cl- (atomic number Z=17)
Solution
1) Na+
A neutral Na atom has an atomic number (Z) 11. In an uncharged Na atom, the atomic number correspond to the number of protons in the nucleus and an equal number of electrons in the outer-nuclear region.
Therefore, the atomic number (Z), the number of protons, and the number of electrons for a Na atom are 11.
The Na+ ion is charged. The positive charge indicates a loss of one electron to give an electron-deficient cation.
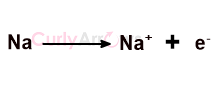
The number of electrons in the Na+ ion is now 10, whereas the proton number is unchanged at 11.
2) Ar
A neutral Ar atom has an atomic number (Z) 18. The atomic number equals the number of protons and electrons in an uncharged atom.
Therefore, the proton and electron number of Ar is 18.
3) Cl-
A neutral Cl atom has an atomic number (Z) 17. The atomic number 17 also indicates 17 protons in the nucleus and 17 electrons in the outer-nuclear region of the Cl atom.
Cl- is a negatively charged anion. The gain of one electron makes a neutral Cl atom attain a negative charge.

Therefore, the electron number is 18 in Cl- and the proton number is 17.
