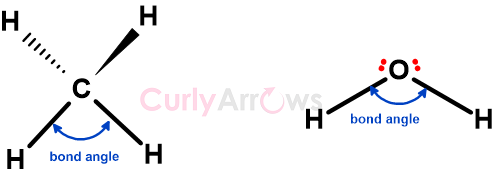
How does molecular geometry or shape influence reactivity?
The arrangement of groups around a central atom creates its three-dimensional shape. And this shape affects how it interacts with other molecules or ions.
The interactions could be bonding - to form new bonds or nonbonding- affecting the physical properties of solubility, melting point, boiling point, polarity, and more.