Types of Solids- Crystalline and Amorphous
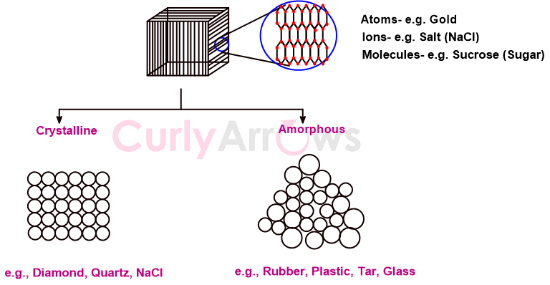
Solids are made up of many particles, and depending on how they are arranged, solids can be divided into crystalline solids and amorphous solids.
The particles may be atoms, ions, or molecules.
For example, gold atoms, NaCl ions, and sucrose molecules compose the solid-state.
Formation and arrangement of solid particles
Suppose a solid is heated to a particularly high temperature, and the molten liquid is gradually cooled. In that case, the constituent particles get sufficient time to arrange themselves into a highly ordered crystalline solid structure, a process called annealing. Annealing effects the crystalline properties of the solid and is sometimes used to rearrange the particles until they reach the crystalline state.
Examples of crystalline solids are Diamond, NaCl, and Quartz.
However, instead of gradually cooling, if the molten liquid is cooled rapidly or if the liquid vapors are frozen suddenly, the particles do not get enough time to organize efficiently. The arrangement of particles is disordered, and the resulting solid is called an amorphous solid.
Examples of amorphous solids are Rubber, Glass, Tar, Plastic, and Quartz glass.
However, the notion of a perfect crystalline solid is false. One can obtain an ideal, perfect crystalline solid at 0K, a very low temperature at which the particles do not have any thermal energy. Therefore, they have no motion; there is no disorder leading to zero entropy. At this temperature, the particles can arrange themselves in perfect order. At all other temperatures, minor irregularities always exist in the crystal structure, and an ideal crystalline solid is not obtained.
Crystalline solid structure
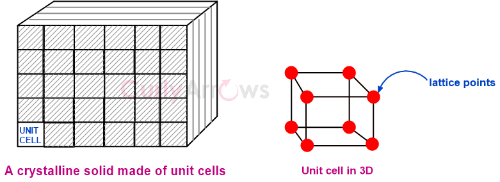
The smallest building block of a crystal is called a unit cell. On the corners of the unit cell, the particles of the solid occupy their positions.
In addition to the corners, some atoms may also be present at the center, on the faces, or at the edges of the unit cell. These places that atoms occupy in a unit cell are called the lattice points.
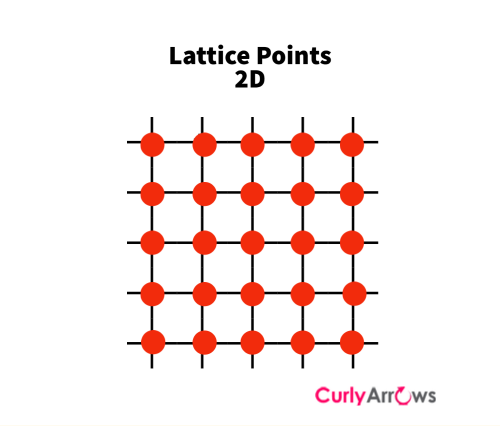
Many such unit cells repeat to give a complete crystalline lattice. It is called a lattice because, in the two dimensions, a crystal looks like a series of points with criss-cross lines like a network or mesh.
When the entire crystal is shown as lines and points in the three dimensions, it's called a crystal lattice. As the unit cells extend in all three dimensions in space, a crystal lattice is also known as a space lattice. These repeating structures of the unit cell in all three dimensions give an entire crystalline solid.
.png)
Therefore, by maintaining the order of the arrangement of the unit cells and their periodic repetition, an entire crystalline solid is obtained.
One way to understand is by comparing the crystalline solid with a human body. The human cell is the basic unit of all the living organisms, like the unit cell is a crystalline solid. The biological cells unite to form a multi-cellular organism. Similarly, many unit cells combine in the three dimensions to give a crystalline solid.
Order of particles in Crystalline solids
Crystalline solids have both short-range and long-range order, which means that the constituent particle arrangement is ordered and regularly repeated in smaller and extended regions. Therefore, one can predict the structure of the entire crystal if the structure of only one unit cell is known.
Amorphous solid structure
The amorphous solid has a tightly packed, random arrangement of the constituent particles. The amorphous solids have no unit cell or periodic, repetitive arrangement of cells. The angles and the lengths between the particles differ.
Order particles in Amorphous solids
Due to their random arrangement of the particles, amorphous solids do not have any long-range order but may have short-range order.
It means that there may exist small, scattered regions in the amorphous solids, which may have an ordered arrangement of the constituent particles as seen in the crystalline solids. Such crystals that are part of an amorphous solid are called crystallites.
.png)