A decomposition reaction breaks down the substrate into two or more smaller, simpler molecules.
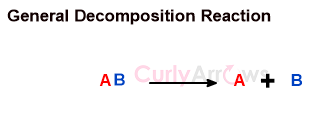
The decomposition reaction is initiated by obtaining energy from heat, light, or electricity sources.
Depending on the source, the decomposition reaction is of three types-
a) Thermal Decomposition
High-temperature conditions cleave the covalent bond to generate smaller, simpler molecules.
For example, carbonic acid bond dissociates at high temperature to give CO2 and H2O molecules.

b) Photochemical Decomposition
Here, light energy is used to cleave the chemical bonds.
The free radical formation reactions proceed under this condition.
For example, the initiation step of a chlorination reaction involves the decomposition of Cl2 molecules into chlorine radicals.

The radicals formed then undergo substitution or addition reactions.
c) Electrochemical Decomposition
The electric current initiates molten compounds' decomposition reaction (electrolysis) to simpler molecules.
For example, water undergoes electrolysis to form gaseous hydrogen and oxygen gas.

