A benzyl, abbreviated as Bn, refers to the phenylmethyl group (C6H5-CH2-), consisting of a benzene ring attached to a methylene (-CH2-) group.
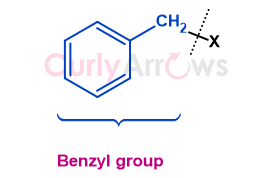
It has a structural formula, C6H5-CH2-X, where the X can be an alkyl/aryl or functional group.
The -CH2- carbon linker connects benzene with the rest of the molecule, called the benzylic carbon, and it is the most reactive position in a molecule, called benzylic position. It is a sp3 tetrahedral methylene carbon connected to a sp2 double-bond benzene carbon.

The hydrogens attached to the benzylic carbon are called benzylic hydrogens.
Significance of the benzylic position
The benzyl group utilizes the benzene ring’s aromatic nature to accommodate and stabilize carbocation, carbanion, and free radical formation and, therefore, is a good reactive intermediate in many organic reactions.
The stabilization is due to the additional delocalization of the positive and negative charges and the radical on the benzene ring carbon atoms.

The delocalization is possible due to the conjugation of the pie bonds of the benzene ring with the p-orbital of the carbocation, carbanion, and free radical. The planarity and the orbital overlap allow for the delocalization or the spread of the charges/radical onto several atoms.

The benzylic hydrogens are more reactive than allylic hydrogens, especially in radical substitution reactions where the benzyl hydrogen is replaced with a halogen, forming benzyl halides.
The benzylic halogenation helps introduce a halogen leaving group where none was present, so a multistep synthesis is now possible.

.png)