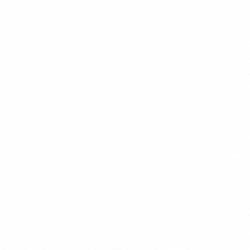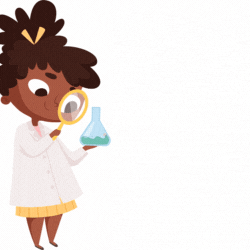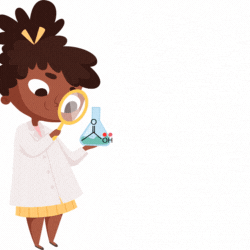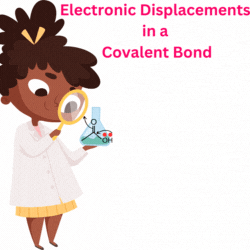An atom or group of atoms that can pull the bond electrons towards itself or push the bond electrons from itself and decreasingly transmit the effect along the sigma (σ) bonds of the carbon chain inducing permanent polarization in the molecule. Such an effect is called the Inductive effect.
The atoms that pull the electrons towards themselves are said to be electron-withdrawing and show a negative inductive (-I) effect. Examples are: -OH, -CN, -NO2, -F, -CHO, etc.
The atoms that push the electrons away from themselves are said to be electron-donating and show a positive inductive (+I) effect. Examples are: -O-, -CH3, -COO-, etc.
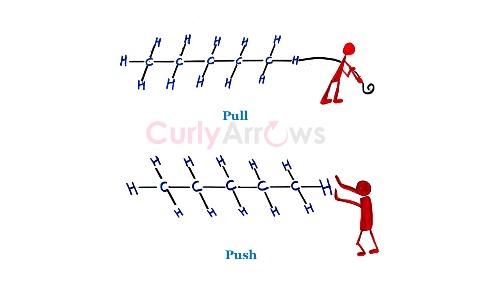
Sometimes, an arrow is drawn on the sigma bond to indicate the direction of pull or the push of the bond electrons.
The arrowhead pointing towards an atom or group of atoms indicates its pull.

The arrowhead pointing away from an atom or group of atoms indicates the push of the bond electrons. In the above example, -NO2 is pulling, and -CH3 is pushing the bond electron density.
Read more about the - Inductive Effect.