Lone pair is a set of electrons present in an atom’s valence shell that did not participate in a covalent bond formation reaction; therefore, they are also called the non-bonding electrons.
While drawing the molecules’ structure, the lone pair electrons on shown as dots (..) above the atom.
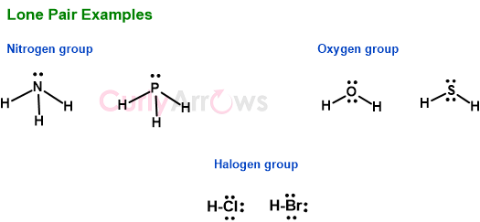
The Nitrogen group carries one lone pair; the Oxygen group has two, and the Halogens have three lone pairs.
The physical properties of shape, H-bonding, polarity, and the dipole moment are affected by the lone pair's presence on an atom.
Lone pairs containing atoms act as donors of electrons and are Lewis bases.
Related Article - Lone pair in Chemistry.
