A Neutron is an uncharged, neutral subatomic particle found in an atom's nucleus, denoted as n or n0.
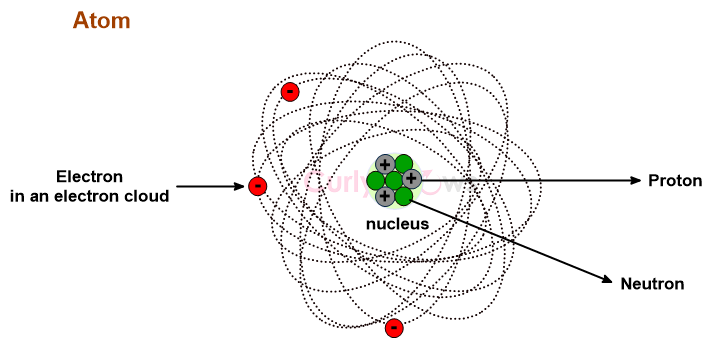
The neutron and positively charged protons constitute the nuclei of all atoms except Hydrogen. The Hydrogen (H) atom's nucleus contains no neutron; however, its isotopes, Deuterium (2H) and Tritium (3H), have one and two neutrons.
The neutron has a mass of 1 AMU, where 1 AMU equals 1.674927 X 10-27 Kg. The neutrons and the protons, with their identical weights, together contribute to 99.94% of the atom's mass.
While the number of neutrons does not affect the element's identity; however, changing the number of neutrons gives various isotopes of the same element.
For example, the Carbon atom has three isotopes - 12C, 13C, and 14C. All three isotopes have 6 protons; however, the number of neutrons is 6 in 12C, 7 in 13C, and 8 in 14C.
Neutrons are essential for maintaining an atom's stability.
