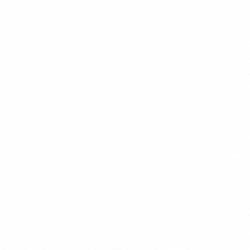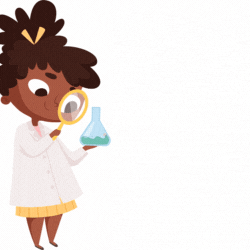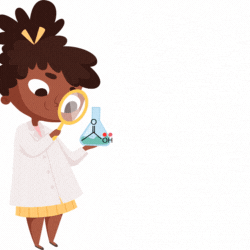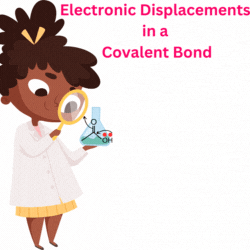Resonance theory explain various observed properties in a molecule using electron delocalization and multiple Lewis structures since a single Lewis structure can only describe one but not all its properties.
The several structures that show delocalization of electrons, mainly pie electrons over a conjugated system, are called the contributing or resonance structures, each separated by a double head arrow. The molecules that require such multiple resonance structures are said to show resonance.

For example, resonance structures 2 and 3 explain the triple and single bond nature of CO2, whereas structure 1 describes its double bond nature.

A resonance hybrid is an average of all resonance structures and is closest to the real nature of the molecule.
Read more about Resonance.