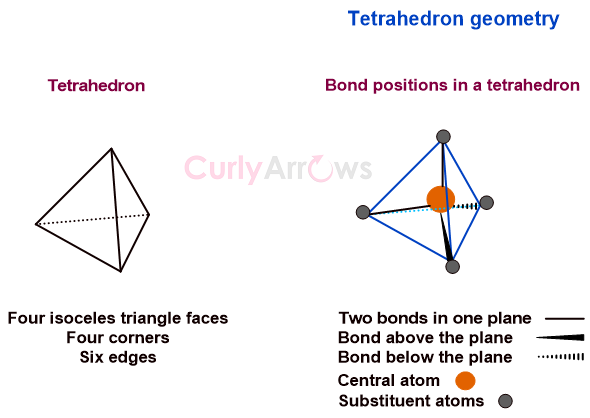
In a regular tetrahedral molecular geometry, a central atom is surrounded by four substituents that occupy the four corners of a tetrahedron. The substituents are called ligands if the central atom is a metal.
However, inorganic compounds like the P4 molecule exist where the four Phosphorus atoms occupy four corners without any central atom.
In a perfect tetrahedral organic molecule like CH4, the central atom of carbon directs the bond to the four corners such that the angle between two adjacent bonds is 109.5o.

In a tetrahedral geometry, two of the four bonds are in one plane; the third is above, and the fourth is below the plane.
(Read how to represent structures in 3D using lines, dash, and solid wedges)

One can imagine a tetrahedral geometry to look like twisting one’s body while standing straight and arms out two legs in one plane, one arm above and another below the plane.

If one or two of the corner substituents is a lone pair, in that case, the bond angle reduces to avoid inter-electronic repulsions. For example, in NH3 and H2O molecules with one and two lone pairs, the angle between the bonds is reduced to 107o and 104.5o from the ideal 109.50. The repulsion also distorted the shape from the ideal tetrahedral in CH4 to pyramidal in NH3 and bent in H2O.
The change in tetrahedral bond angles and shape with respect to the substituent atoms or lone pairs are covered by the Valence shell electron pair repulsion (VSEPR) theory.
VSEPR theory represents a perfect tetrahedral molecule with all bond pairs and no lone pairs as AX4.
Other examples of molecules showing the tetrahedral arrangement of substituents or ligands are- perchlorate ion (ClO4-), Phosphate ion (PO4)-3, ammonium ion (NH4+), saturated compounds of Sn, Ge, and Si like SnH4, SiH4, and GeH4, Ni(CO)4, TiCl4, tetrakis(triphenylphosphine)palladium(0) (Pd[P(C6H5)3]4), etc.
