In a regular trigonal planar molecular geometry, a central atom is surrounded by three equally spaced substituents in one plane, so joining the three corners would give a triangle.
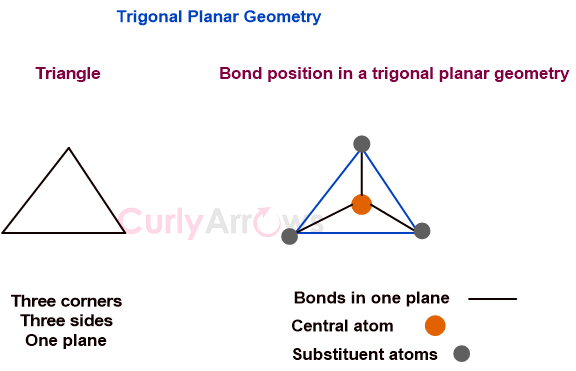
In a perfect trigonal planar molecule like BCl3, the angle between two adjacent bonds intersecting at the central atom is 120o.
The bond angle with respect to the substituents equally spaced is explained using Valence Shell Electron Pair Repulsion (VSEPR) theory that assigns a trigonal planar geometry AX3 indicating that there are three bond pairs and no lone pairs of electrons on the central atom.

Therefore, the central and all three substituent atoms lie in one plane.

A trigonal planar molecular situation can be imagined somewhat like having legs spread out and arms joined so that the ends make three corners of a triangle, all in one plane.

In trigonal planar molecules, the central atom maintains planarity, indicative of its state of hybridization. Hybridization is an event that dictates how and how many of an atom’s orbital would combine to form new bonds.
In trigonal planar molecules, the most preferred hybridization state of the central atom is sp2. In a sp2 hybridization, the central atom has a double bond or an empty p-orbital.
A few examples of molecules with trigonal planar geometry are- BF3, COCl2, AlCl3, SO3, CO32-, NO3-, etc.
