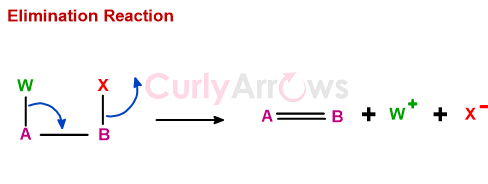
Elimination reaction involves the loss of two groups and the formation of a new bond.
Elimination reaction takes place either by heterolytic or pericyclic mechanism.
In heterolytic elimination, W and X may not leave simultaneously. And after leaving the substrate, the W+ and X- may or may not combine to form one product.
If the two groups are lost from adjacent atoms so that a new double bond (or triple bond) is formed, the reaction is called β (beta) elimination.

The carbon carrying the leaving group (nucleofuge, since it leaves with the bond electron par) is referred to as the α carbon. The carbon that loses the atom as a positive specie is β carbon.
If the two groups are lost from the same atom, it is an α (alpha) elimination. The α elimination forms a carbene, a carbon with 2 bonds and six electrons (or a nitrene, six electrons and one bond on nitrogen).

In a γ (gamma) elimination, the W and X are separated by a carbon atom, and post-elimination, a three-membered ring is formed.

Amongst the three types, β elimination, where the reaction pathway is heterolytic, is the most common. The reactions mainly comprise of dehydrations (elimination of water molecule) and dehydrohalogenations (elimination of Hydrogen halides).
In most β eliminations, new bonds are added to form alkenes (C=C) and alkynes (C≡C). The common β elimination reactions in solution follow three mechanistic routes- E2, E1, E1cb.
E2 mechanism
The E2 reaction occurs in a single step where the two groups depart simultaneously, enabled by the base. Therefore, the two main elements participating in the elimination reaction, and for the reaction to be called E2, are- the base and the substrate.
The base takes away the hydrogen as a proton (H+). The proton’s electrons form part of the new pie bond, an event that triggers the leaving group to leave with its electron pair so that the carbon maintains its tetravalency.

In most E2 elimination, the H and X group (any leaving group) are trans to each other with a dihedral angle of 1800 to allow faster elimination.

E1 mechanism
The E1 mechanism is like E2 in forming the alkene product; however, the two steps occur in reverse.
The first and most important step involves the departure of the leaving group to form a carbocation intermediate. The formation of this intermediate triggers the second step; therefore, the reaction depends mainly on the first step. Since only the substrate is driving the elimination, it is called the E1 mechanism.

In the second step, the proton is picked up by the base, usually a solvent, to form the final alkene.
In the E1 mechanism, the dihedral angle, and therefore, the orientation of the proton to the leaving group is less important than E2. Since in the E1 mechanism, the leaving group is gone before the choice is made as to which direction the new double bond takes place.
However, under E1 conditions, the formation of the most stable, substituted alkene is preferred (Zaitsev’s rule). Therefore, sometimes rearrangement reactions in carbocation intermediates are seen to obtain the stable, substituted alkene as the product. That is an alkene with the largest possible number of alkyl groups on the C=C carbons.

The E2 mechanism is comparable to the SN2 mechanism and competes with it. Similarly, the E1 mechanism competes with the SN1 mechanism.
Elimination reactions are favoured over substitution by increasing the temperature. The elimination reaction has greater changes in bonding where the loss of two single covalent bond forms one pie bond having a greater bond strength.
E1cb mechanism
In the E1 mechanism, X leaves first and then H. In the E2 mechanism, the two groups leave at the same time. E1cb is the third possibility; the H leaves first, followed by the X.
The H leaves as a carbanion; therefore, the cb in E1cb stands for the carbanion mechanism.

E1cb is seen in substrates with poor leaving group X and acidic hydrogen. Acidic hydrogens are identified as those attached to electron-withdrawing groups and, therefore, the easiest to lose as H+.
