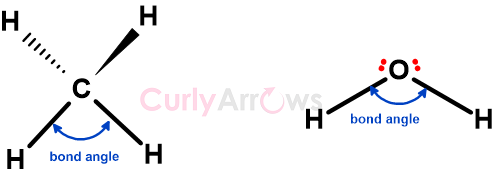
How to determine the hybridization of C in CH4?
Carbon has the atomic number 6, which also equals the number of electrons. According to the rules determining electronic configuration (Aufbau, Pauli, and Hund), these electrons are arranged in the 1s, 2s, and 2p subshells of the Carbon atom to give the complete electronic configuration - 1s2 2s2 2p2. This electronic configuration of Carbon, where the electrons are in their lowest energy states and closest to the nucleus, is called the ground state electronic configuration.