The arrangement of groups around a central atom creates its three-dimensional shape. And this shape affects how it interacts with other molecules or ions.
The interactions could be bonding - to form new bonds or nonbonding- affecting the physical properties of solubility, melting point, boiling point, polarity, and more.
For the sake of reactivity, the molecules prefer a geometry that exposes reactive sites (or the functional group) and does not hinder them. For example, it is easier for a reactive nucleophile to attack a carbonyl carbon in structure A than in B.
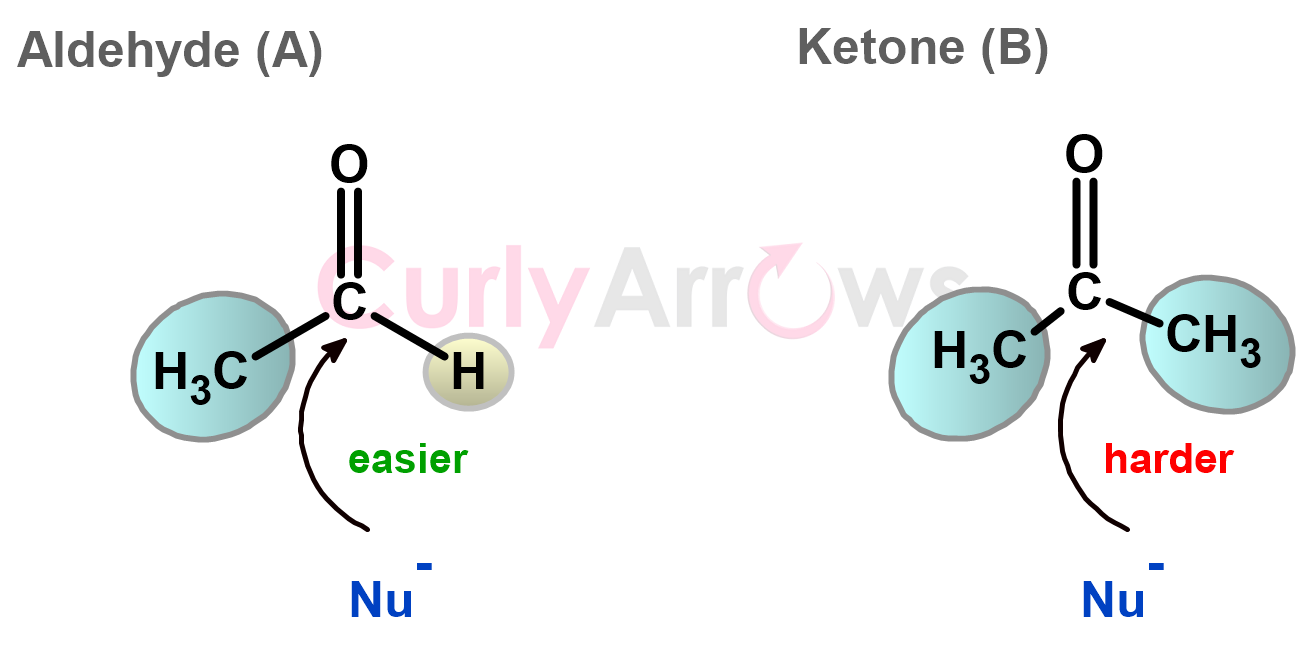
In structure B, the approach of the nucleophile is hindered by the bulk of the ketone. Naturally, it is observed that aldehydes are more reactive than ketones due to the steric ease towards nucleophilic addition reactions.
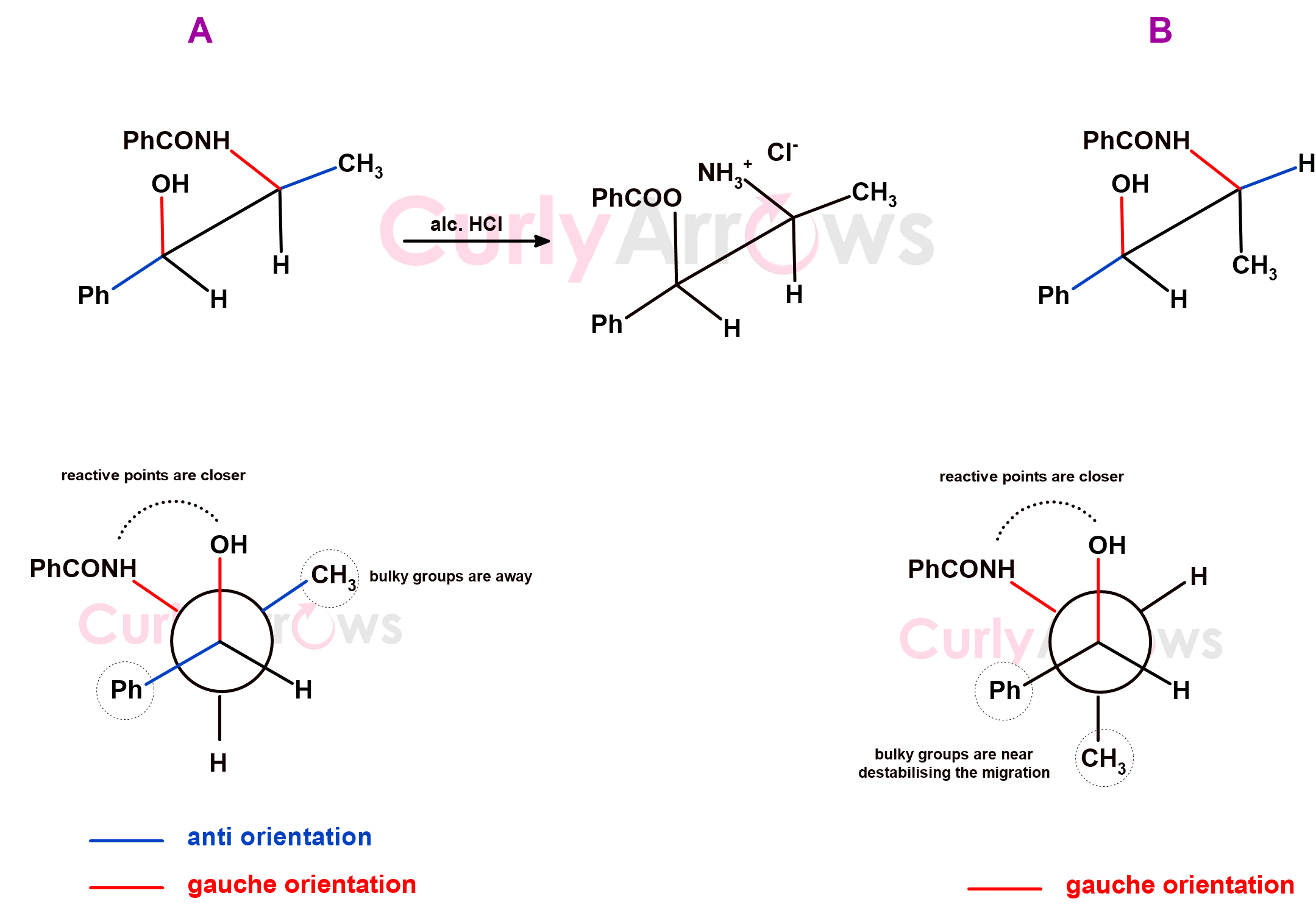
Certain rearrangement reactions fail where the molecule looks similar but has a different conformation. That is, the groups on the atom are the same, but they attach differently, affecting the shape and, therefore, the reactivity.
An example is a rearrangement of N-benzoyl norephedrine, where only one form A undergoes rearrangement by migration from Nitrogen to Oxygen when treated with alcoholic HCl. It only happens when the bulk of CH3 and Ph groups are anti to each other, and Nitrogen is near the Oxygen (gauche to it) so the migration points can engage, react, and form a product.
A few other examples that can be considered based on shape are reactions where adsorption or binding of the reactant molecules encourages chemical reactions, like catalysts. Bulky groups or groups that don’t line up appropriately and hinder the binding of the catalyst to the reactant surface negatively impact the reaction outcome. Enzymatic reactions in our body depend heavily on such binding sites and molecular shapes.
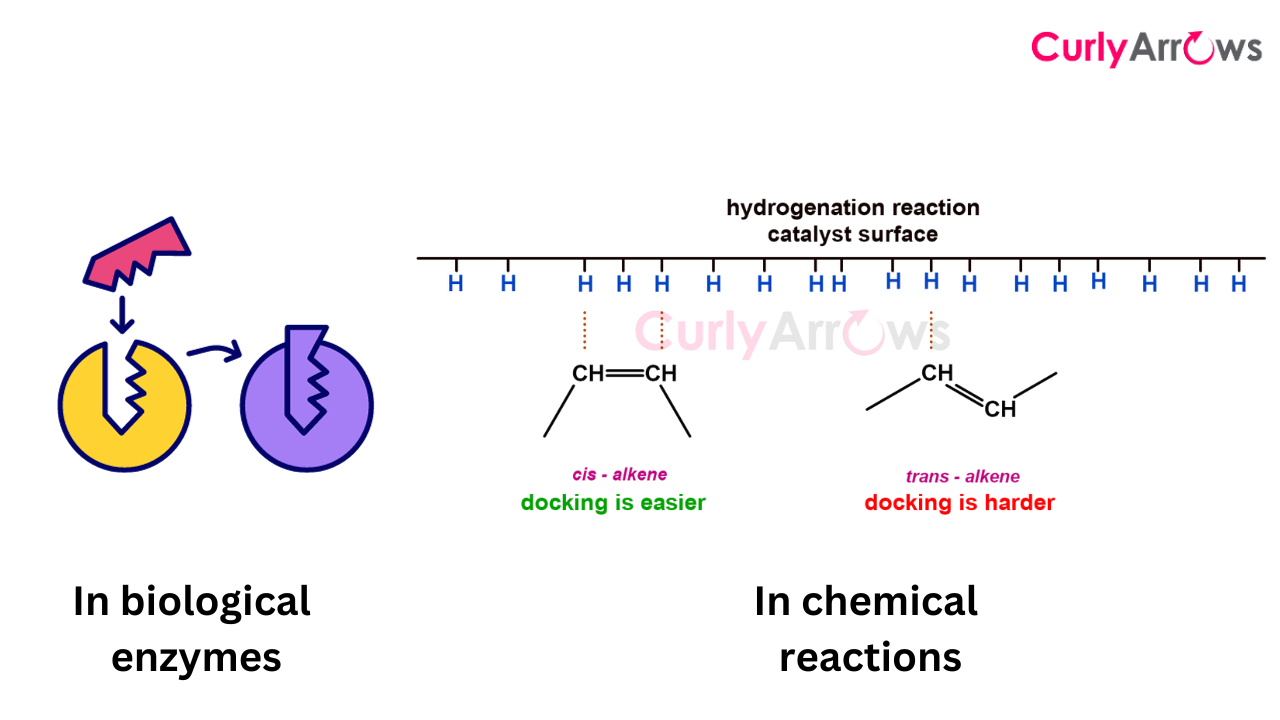
In summary, the shape is crucial to reactivity, affecting how molecules orient and perform under various reaction conditions.
Related Reading –
How does molecular structure influence reactivity?
How molecular structure and functional groups affects physical properties
How central atom creates shapes (VSEPR theory)
