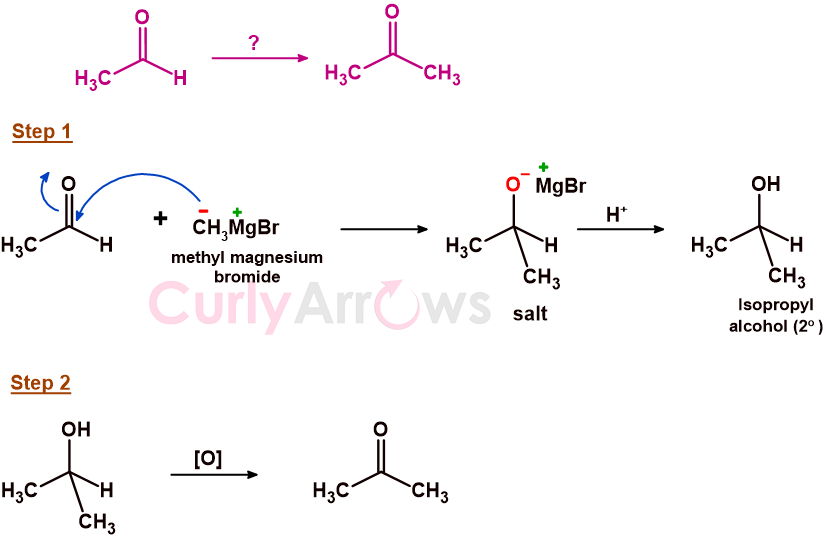
Ethanal is a two-carbon aldehyde, and propanone is a three-carbon ketone. The increase in one Carbon number is possible using a Grignard reagent (Methyl Magnesium Bromide, CH3MgBr) to form a new Carbon-Carbon bond.
In the first step of the reaction, the nucleophile (CH3-) of the Grignard reagent will attack the aldehyde's electrophilic carbonyl carbon (-CHO). The nucleophile's (CH3-) attack will push the pie (π) electrons to the electronegative oxygen and form a negatively charged anion [(CH3)2CH-O-] while still maintaining the tetravalency of the carbon. The negative anion will attract the electropositive magnesium halide such that the charges are balanced, and an ionic salt is formed. The magnesium salt on hydrolysis gives isopropyl alcohol, a 2o alcohol, as the product. Therefore, forming a secondary (2o) alcohol is the first step to conversion to the ketone.
In the final step, the 20-alcohol formed on oxidation with reagents like Na2Cr2O7, CrO3, KMnO4, and PCC gives the desired ketone, propanone, as the final product.