August Kekulé (1866) was the first to propose the alternating single and double bond structure of benzene that can only be possible if carbon could join other carbon atoms and form a ring.
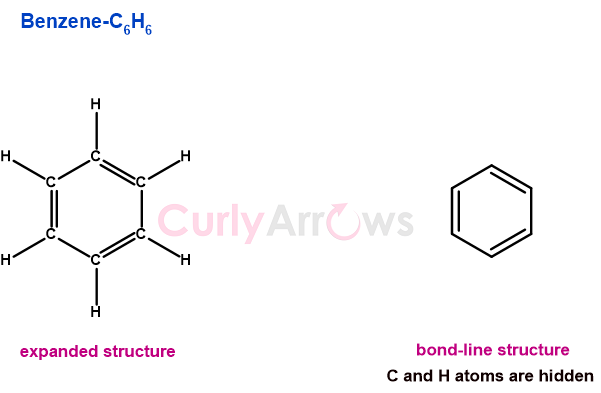
He believed that the tetravalent nature of carbon was responsible for showing catenation, a property of self-linking that allowed carbon to form long chains with itself and other atoms. August Kekulé tried to express this type of bonding by using lines (-) as linkages.
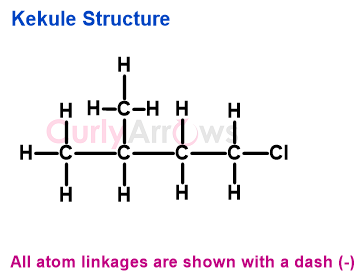
According to him, carbon was the skeleton on which atoms with other valencies could attach and transform further. Kekulé's idea of a carbon-skeleton framework was further used to depict organic structures using zig-zag lines called the bond-line formula and closed ring lines in the polygon formula.
In Kekulé structures, the bonded pair of electrons in covalent bonds are indicated as lines against two dots, as seen in Lewis structures. A Kekulé structure, however, does not show non-bonded electrons or formal charges on the atoms, which is displayed by Lewis structure since electrons were not discovered (1897) until after Kekulé started using this notation.
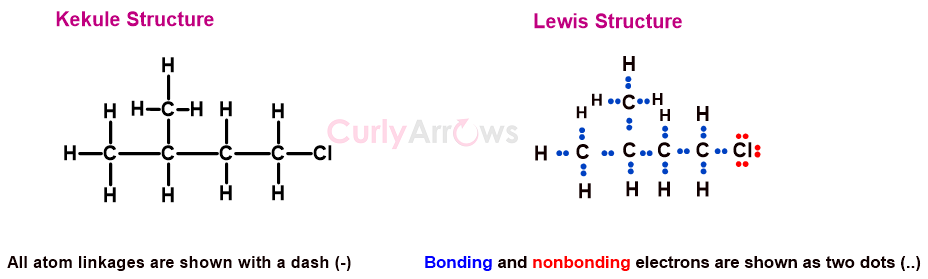
Nowadays, Kekulé's and Lewis's structures are used interchangeably with the non-bonding electrons and formal charges being represented in Kekulé Structures and lines used in Lewis structures for bonds.
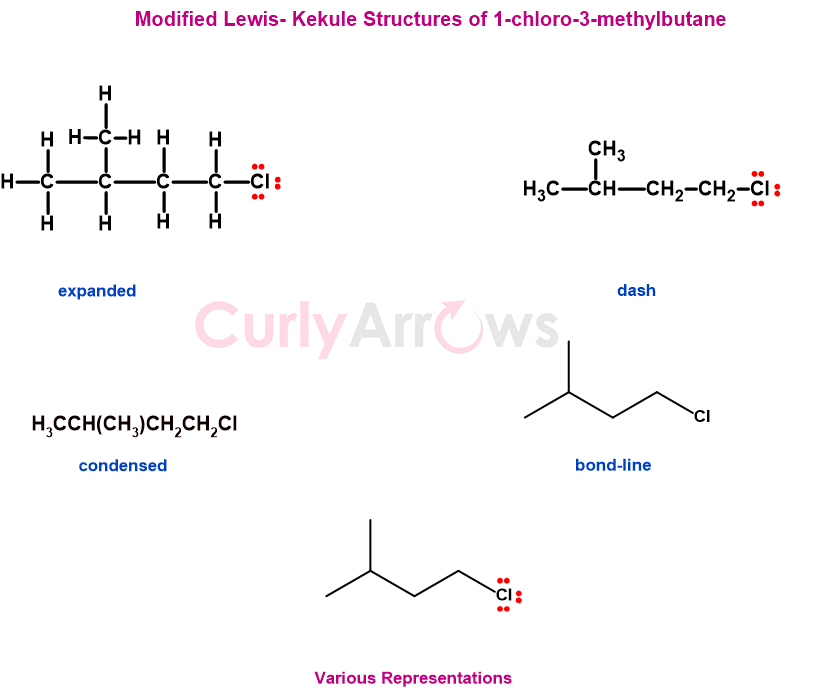
Examples of Kekulé structures
Draw the Kekulé Structures of the following organic compounds
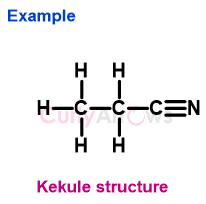
1) Example 1- CH3CH2CN
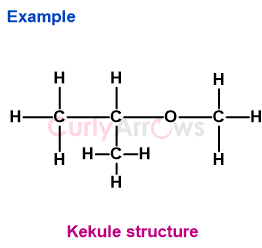
2) Example 2- (CH3)2CH(OCH3)
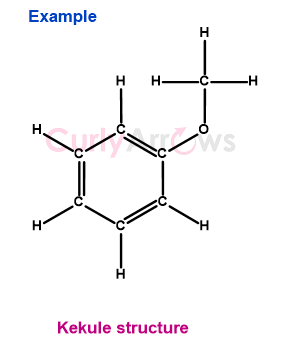
3) Example 3- C6H5OCH3
Drawbacks of Kekulé structural representation
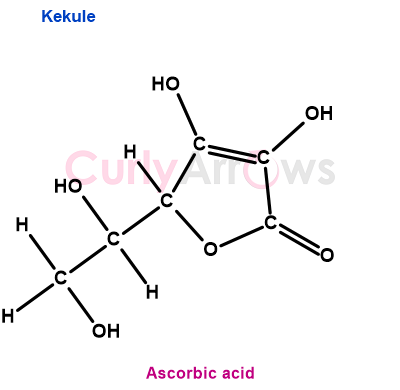
1) Bulky molecules with side chains or substituents are tedious to draw and interpret. For example, ascorbic acid is a bulky molecule that takes up a lot of space, making its drawing time-consuming.
2) Kekulé's style of structural representation does not reveal any information on the molecule's stereochemistry.
