Regioselectivity and Addition to Unsymmetrical Alkenes
Alkenes undergo addition reactions where the substrate (alkene) and the reagent combine to form an adduct. One pie bond of the alkene is sacrificed to create two new stronger sigma bonds with the reagent in the reaction.
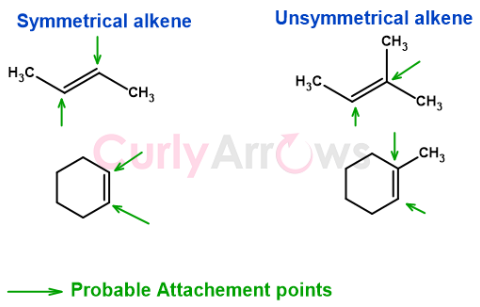
If the alkene is symmetrical with the same number of groups on either end, in that case, the alkene adds to the reagent to form one primary product. However, unsymmetrical alkenes with different groups across the double bond can have more than one way of attachment. This favouritism is called regioselectivity in organic chemistry.
Regioselectivity determines which carbon atom of an unsymmetrical alkene bonds with each fragment of an incoming reagent during this electrophilic addition reaction.
Markovnikov's Rule is a regioselectivity principle in organic chemistry, first proposed by Russian chemist Vladimir Markovnikov in 1869.
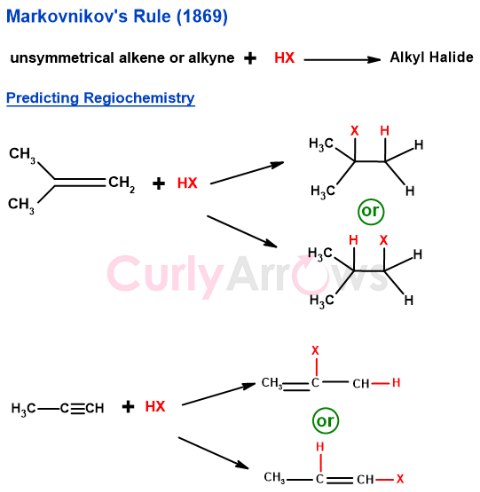
The mystery of the attachment preference was solved by a Russian chemist Vladimir Markovnikov who in 1869 proposed a rule for the addition reactions between unsymmetrical alkenes/ alkynes and hydrogen halides. When an alkene reacts with a hydrogen halide, the two carbons of the double bond are not equivalent — one is more substituted, carrying fewer hydrogen atoms and more carbon substituents, while the other is less substituted. This difference in substitution controls where each part of the reagent attaches, and Markovnikov's Rule is the principle that predicts this outcome reliably.
According to Markovnikov's Rule,
‘In the addition reaction between an unsymmetrical alkene or alkyne and hydrogen halide (HX) to form alkyl halide, the negative part of the reagent (X-) always attaches to the more substituted carbon.’
A mnemonic or shortcut to remember this is-
‘Markovnikov says - NO Member Shall Cheat.'
Where,
NO stands for Negative iOn
Member Shall Cheat for Most Substituted Carbon.

The rule is a direct consequence of carbocation stability as the more substituted carbon produces more substituted carbocation intermediate, as shown in the reaction mechanism below, crucial for the occurence of the addition reaction.
Carbocation Intermediate and Reaction Mechanism
The addition of hydrogen halide (HX) to the alkene double bond is a two-step process via the electrophilic addition mechanism.
The hydrogen halide reversibly exists as H+ and X‑ ions in the reaction. In the first step, the pie bond of the nucleophile alkene picks up the positively charged proton (H+) to give a carbocation intermediate.
The carbocation formed is a stable secondary (2°) carbocation, stabilised by the positive inductive effect — often written as the +I effect — through which neighbouring alkyl groups donate electron density toward the positively charged carbon centre. Therefore, when a double bond picks up a proton, it forms a more stable 2o carbocation at the most substituted carbon than 1o carbocation at least substituted carbon.
This +I effect lowers the energy of the carbocation intermediate, stabilising it and making the more substituted pathway the preferred route of the electrophilic addition reaction.

After the carbocation formation, in the second step of the reaction, the negatively charged halogen attacks the carbocation center to form an alkyl halide. The net result is an exothermic reaction with the loss of one π bond and the formation of two σ bonds, an energetically favorable process.
Hydrogen Halide Reactivity: Bond Polarity and Addition Order
The hydrogen halides (HX) are polar due to the difference in electronegativity. The halogen is more electronegative than the Hydrogen and pulls the bond electrons towards itself. Therefore, the electronegative halogen carries a partial negative charge (δ-) and the Hydrogen a partial positive charge (δ+).
Amongst the hydrogen halides, the reactivity order is-
H-I>H-Br>H-Cl>H-F
The reactivity of HI is highest in the group as it is easy to break the HI bond, due to its long bond length. Post the breakage of the H-I bond, the conjugate ion I- formed is more stable (due to its larger size to charge distribution) and nucleophilic than conjugate ions of HBr, HCl, and HF. The reactivity is least for HF.

Markovnikov's Rule Examples
Equally Substituted Unsymmetrical Alkenes
In an unsymmetrical alkene, if the two carbon atoms of the double bond are equally substituted with the hydrogens, in that case, the halogen would attach to both ends in equal probability to form two products in equal ratios.
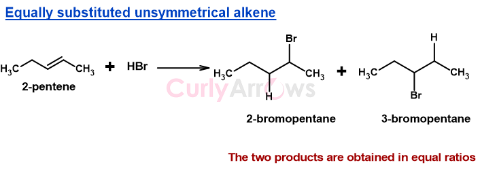
This is because when both carbons of the double bond carry the same degree of substitution, neither carbocation intermediate is more stable than the other, so Markovnikov's Rule has no basis for selecting one carbon over the other. Both addition products form in roughly equal amounts.
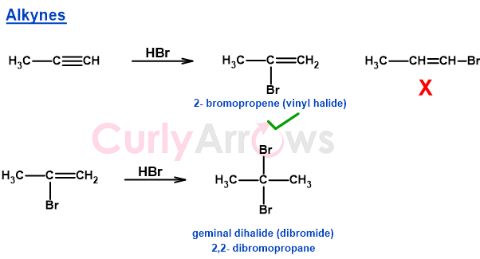
Markovnikov's Rule with Alkynes
The addition reactions of alkynes and hydrogen halide give vinyl halides. In vinyl halides, the halogen is attached to the double bond. If an excess of Hydrogen halide is used, the second HX addition generates geminal dihalides where two halogens are connected to the same carbon atom. The mechanism proceeds like an alkene addition reaction with the formation of a stable carbocation followed by the attack of the bromide ion.
HBr Addition Examples: Propene, Methylpropene, and Butene
The following examples show how Markovnikov's Rule operates across structurally varied alkenes during HBr addition. We see how in each case the difference in carbocation stability at the two double-bond carbons directs the bromide to the more substituted position. That is, the Carbon carrying fewer Hydrogens across the double bond.
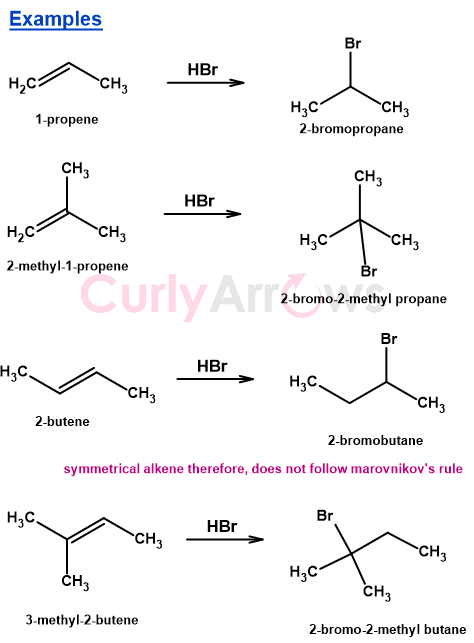
In 1-Propene addition of HBr gives 2-bromopropane.
2-methyl-prop-1-ene on the addition of HBr gives 2-bromo-2-methylpropane.
In example 3, the 2- butene is a symmetrical alkene. It does not follow Markovnikov’s rule for the addition of HBr across the double bond. The addition of Br- from either end would give the same product.
In example 4, the symmetrical nature of 2-butene is lost on substituting a methyl at the 3rd position to give 3-methyl-but-2-ene. It now obeys Markovnikov’s rule to form 2-bromo-2-methylbutane.
The same regioselectivity principle extends beyond HBr. A range of other reagents — including water (H₂O), halogens (X₂), hypohalous acid (HOX), sulfuric acid (H₂SO₄), and iodine monochloride (ICl) — follow Markovnikov selectivity when added across an unsymmetrical alkene, with the electronegative fragment consistently directed to the more substituted carbon.
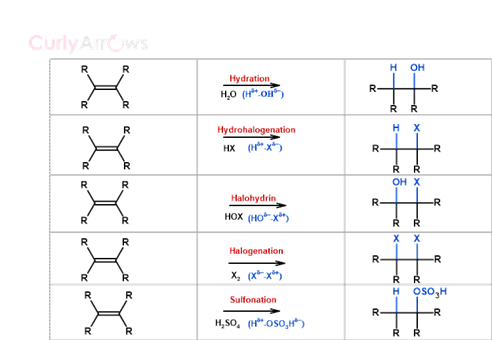
Across all these reagent types, the underlying principle of Markovnikov's Rule remains consistent — the electronegative fragment always attaches to the carbon best able to stabilise the developing positive charge in the transition state.
Anti-Markovnikov Addition and the Peroxide Effect
Anti-Markovnikov addition describes a class of reactions where the halide attaches to the less substituted carbon of an unsymmetrical alkene — the opposite of what Markovnikov's Rule predicts. The most important example is the addition of HBr in the presence of peroxides, a reaction sometimes called the Kharasch effect after Morris Kharasch, who investigated it systematically in the 1930s.
The key difference lies in the mechanism. Markovnikov addition proceeds by an ionic electrophilic mechanism through a carbocation intermediate. Anti-Markovnikov addition with peroxides proceeds by a free radical chain mechanism. Peroxides decompose under heat or light to generate bromine radicals (Br•), which add to the less substituted carbon of the alkene to form the more stable carbon radical intermediate. The resulting product is the anti-Markovnikov alkyl bromide.
It is important to note that the peroxide effect applies specifically to HBr. HCl and HI do not undergo anti-Markovnikov addition under radical conditions because the bond energies involved make the chain mechanism thermodynamically unfavourable for these hydrogen halides. When no peroxide is present, HBr reverts to standard Markovnikov addition through the electrophilic ionic pathway.
Conclusion — Markovnikov's Rule and Regioselectivity in Organic Chemistry
Markovnikov's Rule governs regioselectivity in electrophilic addition reactions. By predicting which carbon of an unsymmetrical alkene receives the halide during addition of a hydrogen halide, the rule allows chemists to anticipate and design reactions with higher confidence. The stability of the carbocation intermediate is the main reason behind the rule — more substituted carbocations are stabilised by the +I inductive effect of neighbouring alkyl groups, directing the halide to the more substituted position.
Understanding Markovnikov's Rule also requires understanding when it does not apply. Anti-Markovnikov addition, driven by the peroxide effect and a free radical mechanism rather than an ionic pathway, reverses the regiochemical outcome for HBr. Together, Markovnikov addition and anti-Markovnikov addition give synthetic chemists complementary tools to place a halide on either carbon of a double bond selectively. Together they build the foundation for understanding broader electrophilic addition reactions across alkenes, alkynes, and beyond.
Frequently Asked Questions
Markovnikov's Rule predicts where each part of a reagent attaches when a hydrogen halide (HX) adds to an unsymmetrical alkene. The hydrogen attaches to the carbon that already carries more hydrogens, and the halide attaches to the more substituted carbon — the one bonded to more carbon groups.
A popular student shorthand: "the rich get richer."
The rule works because of carbocation stability. When H⁺ adds to one carbon of the double bond, a positively charged carbocation forms on the other. The more substituted that carbocation is, the more stable it becomes — neighbouring alkyl groups donate electron density through the inductive effect (+I effect) and hyperconjugation.
The reaction passes through the most stable carbocation intermediate, so the halide ends up on that carbon as the major product.
Anti-Markovnikov addition is when the halide attaches to the less substituted carbon — the opposite of Markovnikov's prediction. The most common example is HBr reacting with an alkene in the presence of peroxides (the peroxide effect, also called the Kharasch effect).
Peroxides generate bromine radicals rather than carbocation intermediates, switching the mechanism from ionic to free-radical. The bromine radical adds to the less substituted carbon because that produces the more stable carbon radical intermediate.
The peroxide effect applies specifically to HBr only. HCl and HI do not undergo anti-Markovnikov addition under radical conditions.
For HCl, the chain propagation step is not thermodynamically favourable. For HI, the H–I bond is too strong for the equivalent radical step to proceed efficiently. Only HBr sits in the energetically favourable window for the free-radical chain mechanism to operate.
Yes. When a hydrogen halide adds to a terminal alkyne, Markovnikov's Rule predicts that H adds to the terminal carbon and the halide adds to the internal (more substituted) carbon, producing a vinyl halide.
If excess HX is used, a second addition follows the same rule, placing both halogens on the same carbon to give a geminal dihalide.
Markovnikov's Rule does not apply to symmetrical alkenes because both carbons of the double bond are equally substituted. There is no preferred carbon for the halide to attach to, so both possible products form in roughly equal amounts.
The rule only has predictive value when the two carbons differ in their degree of substitution.
Yes. Acid-catalysed hydration and oxymercuration-demercuration both follow Markovnikov selectivity, placing the –OH group on the more substituted carbon.
Hydroboration-oxidation, by contrast, delivers the –OH to the less substituted carbon, making it an anti-Markovnikov hydration. Students often confuse these three because all of them produce an alcohol from an alkene.
Yes — a small amount of the anti-Markovnikov product always forms because the less stable (less substituted) carbocation still exists in small concentrations and can proceed to product.
The typical ratio is roughly 80:20 in favour of the Markovnikov product, though this varies with substrate and conditions. The anti-Markovnikov product is considered the minor product under standard ionic conditions without peroxides.
- "The rich get richer" — the carbon with more hydrogens gains another one.
- "H goes to H" — hydrogen adds to the carbon already carrying more hydrogens.
- "No Member Shall Cheat" — Negative Ion → Most Substituted Carbon.
The most reliable shortcut: ask which carbocation would be most stable — the halide attacks there. No memorisation required.
Markovnikov's Rule underpins several areas of applied chemistry:
- Predicting regiochemistry in industrial alkene hydrohalogenation processes.
- Designing pharmaceutical synthesis routes where a halide or hydroxyl must land on a specific carbon.
- Guiding polymer chemistry, where monomer addition selectivity affects the properties of the final material.
Understanding both Markovnikov and anti-Markovnikov pathways gives synthetic chemists two complementary tools for placing a halide on either carbon of a double bond selectively.
