Pre-requisite: Nuclear charge, Valence electrons
Stovetop cooking routinely requires cast iron or aluminium pans but never a glass pan or a pan made of silicon. That’s because the glass pan will shatter at high temperatures, and the silicon pan will burn away.
On the other hand, iron or aluminium pans and metal alloys like steel can withstand and conduct high heat. In addition to heat, such metals can conduct electricity - for example, copper metals in electric wires and tungsten filament in lamps.

Each metal has a unique nature - Iron and Aluminium are hard, but Sodium, Mercury, and Gold are soft. Their special nature allows the metals to be moulded into shapes, beaten into sheets, and drawn into wires like in gold jewellery.
Metals have shine, some are heavy, and few in an alloy form display excellent strength to bear the load and not fracture under stress. Therefore, they find wide applications in building bridges, railroads, cranes, etc.

The properties that make metals so unique lie in how the atoms inside the metal are bonded.
What is Metallic Bonding?
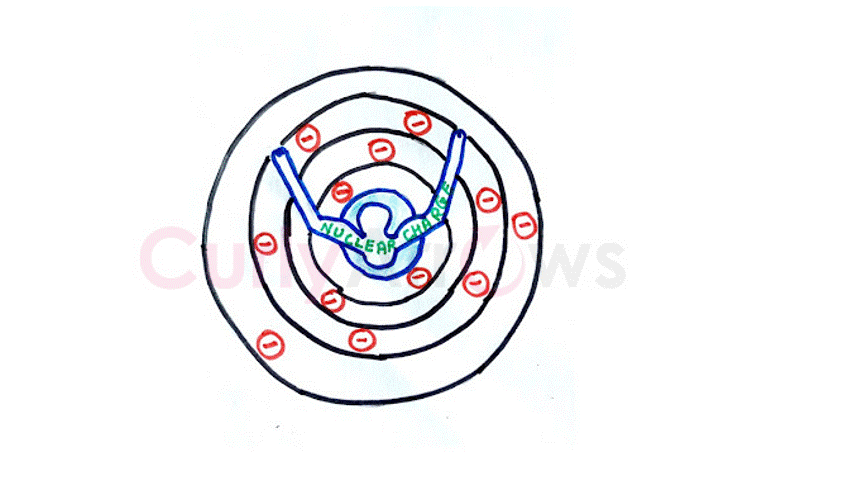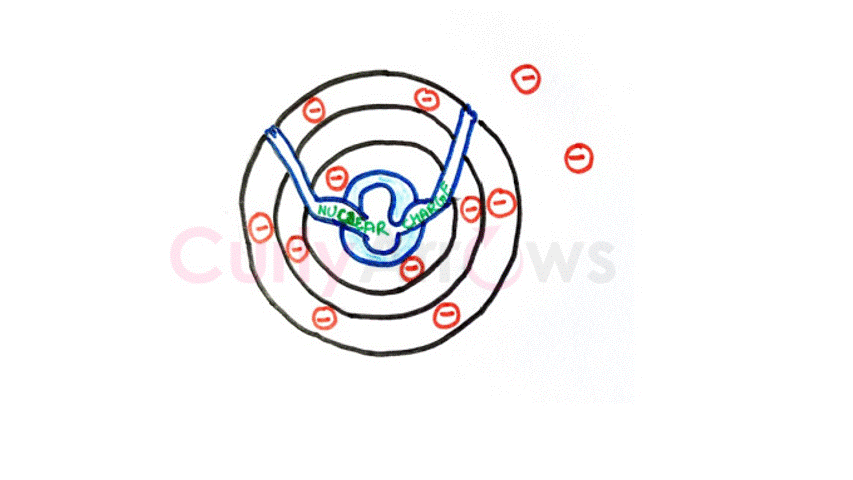
Metallic bonding is the electrostatic force holding a vast collection of metal atoms in a solid structure.
In metals, the atom's size is huge such that the atom's nucleus cannot firmly hold the outermost valence electrons by attraction. The electrons escape from the nuclear hold and drift.
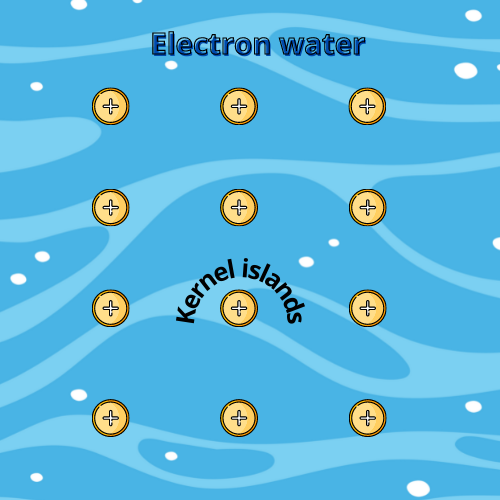
The atom becomes a positively charged kernel on losing its electron. These kernels are spaced at equal distances to avoid repulsion, looking like lattices, and holding the solid structure.
The lost electrons flow through these positive lattices due to electrostatic attraction so that the electrical neutrality of the structure is maintained, like a sea floating around the kernel islands.
Kernels are not ions, as the lost electrons are always replaced in the desired number by the other floating electrons from the nearby atoms. The kernel-valence electrons' relationship is of attraction that does not require a tight binding to an individual atom.
.gif)
The metals ability to easily ionize or lose its electrons is due to its low ionization potential. Also, the metals prefer to undergo electron loss to obtain the stable electronic configuration of the nearest inert gas.
Importance of Metallic Bonding
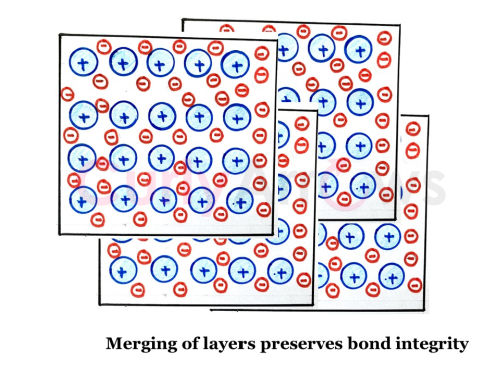
The non-fixated kernel-electron attractive bonding allows the layers of metal atoms to slide past one another and get reorganized without losing identity.
The readjustment allows the metal atoms to merge between layers or to spread into wires, largely contributing to the malleability and ductility.
The free-electron movement also discourages the positively charged kernels from coming too close and showing repulsion, thereby balancing the positive and negatively charged metal structure.
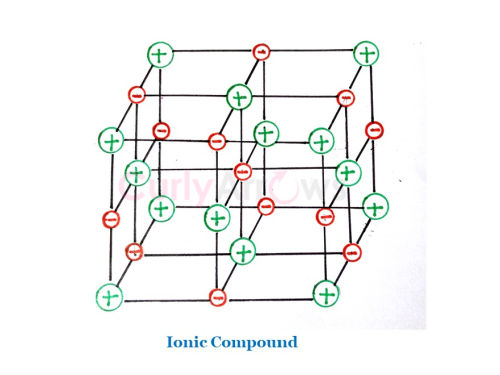
How is metallic bonding different from ionic bonding?
The ionic compounds are formed when metals lose their electrons and nonmetals gain to form ions. However, in ionic compounds, the ions are held in fixed geometry, and in the solid state, the ions are immobile.
Also, the ionic compounds do not show a metal’s unique property of layers sliding past each other.
What is the impact of metallic bonding on the physical properties?
Metals with more valence electrons will have greater mobile electrons, which will raise a metal's conductivity to heat and electricity.
Also, when a metal employs a larger set of electrons for delocalization, subsequent nuclear charge increases, raising the kernel-electron attractive power. The higher kernel-electron attractive strength raises the physical properties of the melting and the boiling point.
For example, Na metal has one valence electron, and Al has three valence electrons. Na can lose one electron to give Na+ kernel and one free e-. By losing the electron, the nuclear charge of Na is +1. If Al loses its three mobile electrons (e-) to form the Al3+ kernel, it will have a nuclear charge of +3.
The higher the nuclear charge, the higher the kernel-electron attractive power will be. Therefore, Al3+ will show higher attractive strength for the mobile electrons than the Na+ kernel raising the melting and the boiling point.
| Element | Melting Point | Boiling point | Valence Electrons |
|---|---|---|---|
| Sodium | 97.79 oC | 882.9 oC | 1 |
| Aluminium | 660 oC | 2470 oC | 3 |
The transition metals with greater free electrons in the 3d and 4s orbitals than s-block or p-block metals will show higher melting and boiling points.
| Element | Melting Point (oC) | Boiling Point (oC) | Valence electrons |
|---|---|---|---|
| Sodium (s-block) | 97.79 | 882.9 | 1 |
| Magnesium (p-block) | 650 |
1091
| 2 |
| Aluminium (p-block) | 660 | 2470 | 3 |
| Iron (transition, d-block) | 1204 | 2750 | 8 |
| Nickel (transition, d-block) | 1453 | 2732 | 10 |
Increase in Valence e – increases M.P., and B.P.
Note that the melting points are lower than the boiling points as a metal's bonds are only loosened on melting. However, boiling a metal requires higher energy input, breaking the kernel-electron attractive force and truly destroying the crystal structure.
What are the applications of metallic bonding?
The property of metallic bonding is harnessed for thermal, electrical conductivities, lustre, and shape. Also, mixing different metals forms alloys with new separate improved identities that display greater potential and have broader applications.
For example, steel, an alloy of iron and carbon, is harder than iron itself.
An alloy of Nickle and titanium is called nitinol, responsible for shape memory and capable of springing back to shape and displaying superelasticity at different temperatures.
The science of metallic bonding is also used to tweak the crystal structure of semiconductors (Si, Ge, etc.) to introduce n-type (negative type) or p-type (positive type) defects. The defect dramatically improves the semiconductor's processing power and performance, which finds broader applications in transistors, diodes, circuits, computers, cars, and smartphones.