Alkene is a double bond containing hydrocarbon compound. Since the hydrocarbon compounds can be short or long chained, the double bond's position could be anywhere along the hydrocarbon chain.
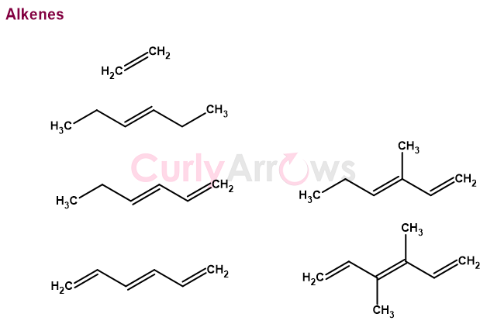
The double bond's position on an alkene can reveal different substitution patterns- mono, di, tri, tetra substituted alkenes.
The substitution pattern can control the regiochemistry of an addition reaction, a reagent's decision on which side of the alkene it should add to form an addition product.
Difference in reaction between symmetrical and unsymmetrical alkene
In symmetrically substituted alkene, the reagent has an equal probability of attacking from either end of the alkene to form one product.
However, for an unequally substituted alkene, the reagent can attack either side of the double bond that can give more than one product, one of which will be more dominant than the others. Still, it is observed that the reagent always attacks the most substituted carbon.
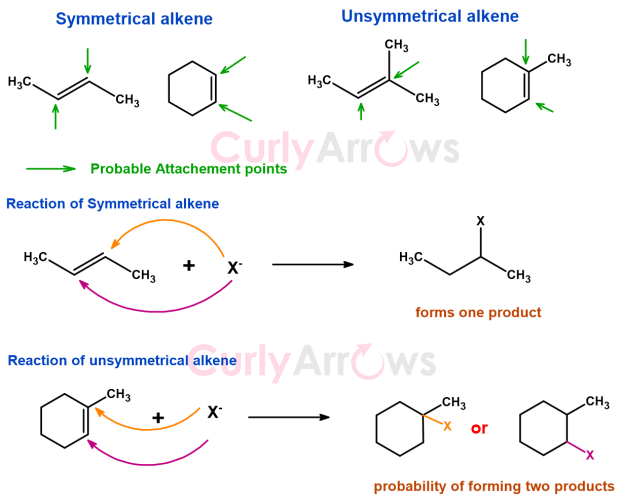
Understanding substitution pattern is very important to preconceive the product formation in an alkene addition reaction. Also, in reactions where alkenes are the product, chemistry always favors the formation of most substituted alkene.
Therefore, it is essential to understand -
a) What is the most and least substituted alkene? and
b) How to identify the most and least substituted alkene?
Alkene Substitution Patterns –
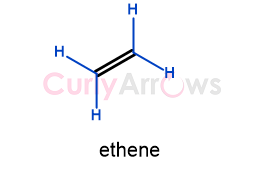
a) Unsubstituted Alkene
An unsubstituted alkene is the simplest alkene. Between two carbon atoms, there is only one double bond present. It has no carbon neighbors, that is, any alkyl groups attached to the double bond. Ethene is an example of an unsubstituted alkene.
b) Substituted alkene
The alkene double bonds with alkyl (carbon) neighbors are substituted alkenes. They are called substituted because the hydrogens of the alkene double bond are replaced with alkyl groups.
The substituted alkene comprises four types (for each of the hydrogens replaced) - Monosubstituted, Disubstituted, Trisubstituted, and Tetrasubstituted Alkene.
Mono, Di, Tri, Tetrasubstituted Alkene
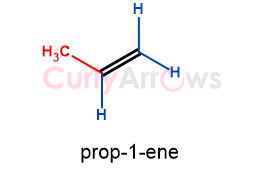
When one end of an alkene has one carbon neighbor, it is called monosubstituted alkene. In monosubstituted alkene, one of its hydrogens is substituted with an alkyl group.
For example, if one hydrogen of ethene is substituted with methyl, prop-1-ene is obtained, which is a monosubstituted alkene.
When two hydrogens of an unsubstituted alkene are replaced with two alkyl groups, the alkene is disubstituted. The two alkyl groups can be on the same side or the opposite.
Similarly, replacing three and four hydrogens of an unsubstituted alkene gives tri and tetrasubstituted alkene.

The disubstituted alkene can have alkyl groups arranged in trans (opposite), cis (same side), and geminal (on one carbon atom) positions.
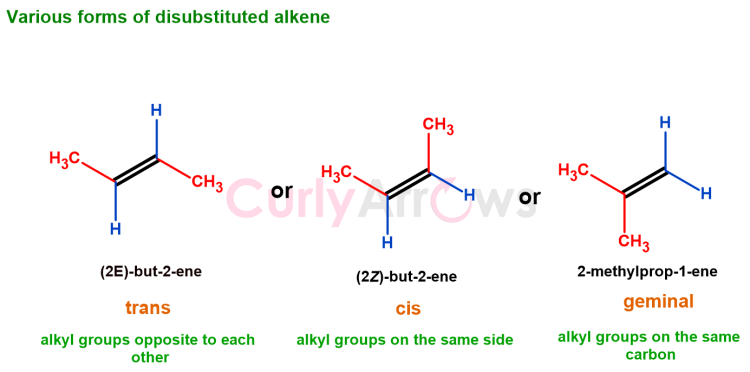
Therefore, the most substituted alkene will have more carbon neighbors as alkyl groups. The least substituted alkene will have the least carbon neighbors and more hydrogen atoms.
Identifying the Most substituted alkene
In a long chain alkene hydrocarbon, to understand which alkene is most and least substituted, let’s look at the following example of (3E,5E)-5,6,8-trimethylnona-1,3,5,7-tetraene. The given molecule is a nine-carbon chain with the four alkenes at 1, 3, 5, and 7 positions.

On counting from the left to the right, alkene 1 has only one alkyl substituent at carbon 2 (carbon 2-3 bond) and is a monosubstituted alkene.
Alkene 2 is disubstituted (carbon 3 and 4 substituted), whereas alkene 3 between carbon 5 and 6 is tetrasubstituted. All the hydrogens of the alkene 3 (between carbon 5 and 6) are replaced with alkyl groups.
The fourth alkene at carbon 7 is trisubstituted. The carbon atom at the 7th position still has one hydrogen intact and unsubstituted. Therefore, amongst the four alkenes, the tetrasubstituted alkene 3 (at C5-C6) is the most substituted, and the alkene 1 (at C1-C2) is the least substituted.
The most substituted alkene carbon atom
The most substituted carbon atom of an alkene is the one that has lost both the hydrogens to an alkyl group. For example, structures 1 and 2 are disubstituted alkene, but the carbon atom (carbon 1) of structure 2 is the most substituted.
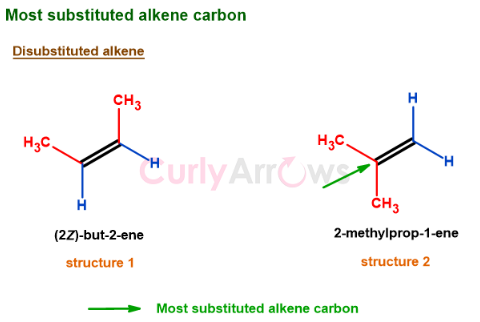
Next: Which side of an alkene does the reagent attack and why?
Also Read : Stability Order of Substituted alkenes versus unsubstituted alkene
