Learning Objective: To study the octet rule by G.N. Lewis, considering both ionic and covalent bonding, with examples.
Skill Level – Intermediate
Prerequisites:
Related –
Chapter: Bonding in Atoms
Author's Note: In the previous chapter, we discussed the difference between valence and core electrons and how to write the electronic configuration.
In this chapter, we will start learning about how various atoms form bonds. For that, we must know how atoms differ from each other and what they pursue while bonding.
G.N. Lewis postulated an octet rule that proposed why atoms primarily form bonds. We will see two types of chemical bond formation between atoms: ionic and covalent, both of which fulfill the octet rule.
Octet Rule in Chemistry - Ionic and Covalent Bonding
As early as 1916, G.N. Lewis knew that in a chemical reaction, the central nucleus of an atom remains unaltered. However, it is the electrons in the outer nuclear region of atoms that participate in chemical reactions, driving transformations.
Still, only some atoms participate, and others don’t.
He assumed that all noble gases (He, Ne, Ar, Kr, Xe, Rn) are stable, and that their electron configurations are what guarantee their stability and chemical inertness. According to his hypothesis, their filled outer-nuclear shell, which contains eight electrons, gives them that stability.
Therefore, Gilbert N. Lewis formulated an ‘octet rule’ that says that the number 8 is the stable outer electronic configuration that the atoms pursue. And to attain the octet outer-electron configuration of the nearest noble gas, atoms gain, share, and lose electrons and therefore participate in covalent bonding.
An exception is Helium, which prefers the duplet configuration rather than the octet state.
Achieving octet by losing or gaining electrons - Ionic Bonding
The atoms can attain a stable electronic configuration of their nearest inert gas by losing or gaining electrons.
.png)
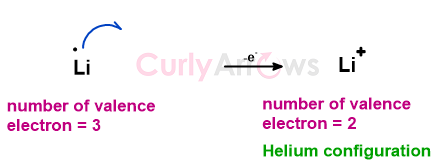
For example, lithium has one electron (denoted by a dot.) more than the inert gas Helium, which is its nearest inert gas. By losing one electron, it will form a Li+ ion (cation) with an inert gas configuration of Helium.
The Fluorine atom that belongs to the same row of the periodic table as Lithium has seven electrons more than Helium or one electron less by Neon.
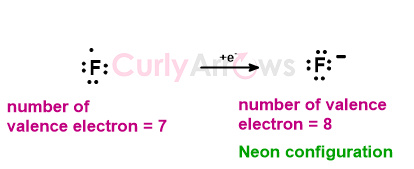
Instead of losing all 7 electrons to reach the Helium configuration that would require very high energy, the Fluorine atom prefers to gain one and attain the electronic configuration of Neon. By gaining one electron, the Fluorine atom forms an F- ion (anion) with Neon's inert gas configuration.
When the atoms lose and gain to each other, it gives them a chance to form an ionic bond.

Therefore, when one excess electron of Li is accepted by F, an ionic bond between oppositely charged Li+ and F- ions forms to give LiF, an ionic compound.
Achieving octet by losing or gaining electrons- Covalent Bonding
The atoms that don’t prefer to lose or gain electrons take a middle path: sharing electrons and forming a covalent bond.
For example, Carbon, Oxygen, and Nitrogen atoms have 4, 5, and 6 outer electrons. They require 4, 3, and 2 more electrons to reach an octet, and they do so by forming covalent bonds.
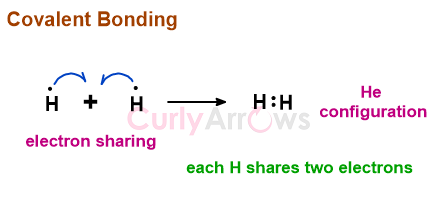
A covalent bond is a chemical bond formed between two atoms by sharing their outer valence electrons. By sharing, both the atoms of a covalent bond attain the nearest inert gas configuration.
For example, the Hydrogen atom needs a second electron to achieve a noble gas configuration of Helium. Therefore, when two hydrogen atoms unite, they share their two electrons to form a bond such that each atom has two electrons in its valence shell.
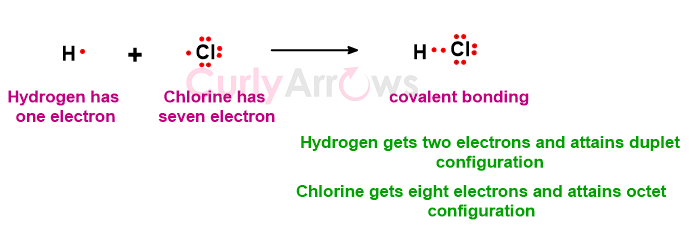
Other combinations are the H atom (less by one electron) with the Cl atom (less by one electron) to form an HCl molecule where H attains duplet, and Cl attains octet configuration.
C atom (less by four electrons) and four H atoms (each less by one electron) combine to form CH4 where C attains octet and H attains duplet configuration.
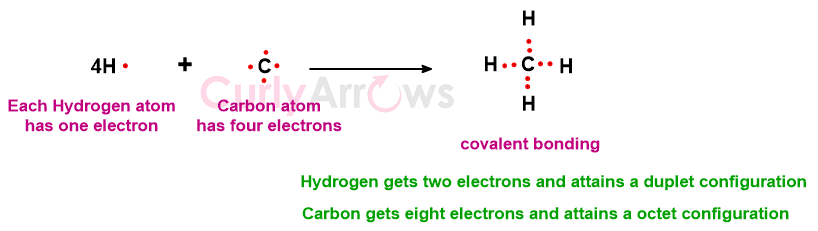
N atom (less by three electrons) and three H atoms (each less by one electron) combine to form an ammonia (NH3) molecule.
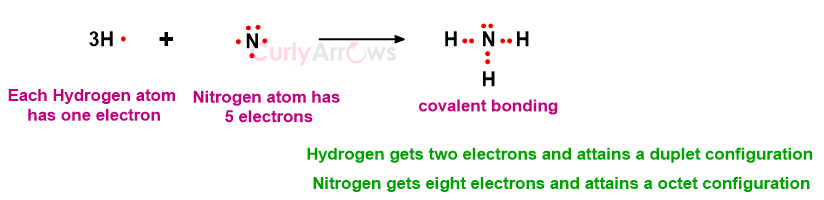
The structures in which dots symbolize covalent bonding are called Lewis structures. In a Lewis structure, each valence electron is shown as a dot.
The shared electrons that form part of the covalent bond are called bond pairs. Sometimes, the bonding pair of electrons is represented by a dash (-) rather than dots when drawing a molecular structure. (The different molecular structure drawing styles are part of the chapter - Drawing Organic Structures)
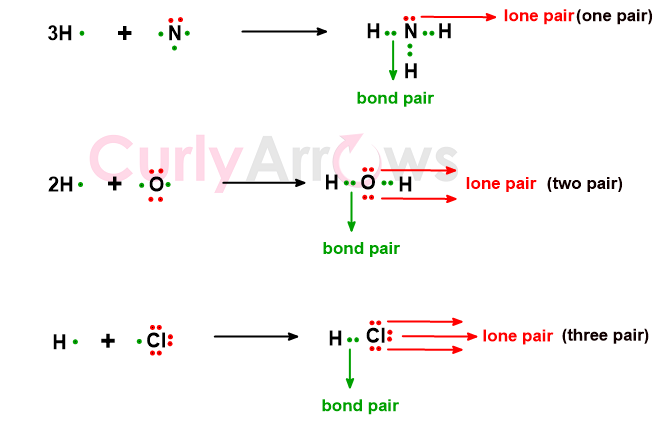
The electrons on an atom that refrained from participating in the sharing and subsequent covalent bond formation are called the non-bonding electrons. They still are, however, part of an atom’s octet. A non-bonding electron, also known as a lone pair, is denoted by two dots (..) on the atom.
For example, Nitrogen only uses three of its five electrons for covalent bonding to form an NH3 molecule. Oxygen used only two of its six electrons to form one water molecule, whereas chlorine only used one of its seven electrons to form HCl. The unused electron pairs on Nitrogen (one), Oxygen (two), and Chlorine/Halogen (three) are its lone pairs.
Subscribe to the study program on Organic Chemistry Fundamentals.
Next: Limitations of the Octet Rule
