Understanding the difference in Oxidation-Reduction in Organic versus Inorganic Chemistry
Inorganic chemistry describes oxidation in two ways. The first method is through the loss of electrons, and the second is by the increase in oxidation number.
An example of the first method is when Sodium loses an electron (undergoes oxidation), and the lost electron is picked up by Chlorine (undergoes reduction) to form the final product. The formation of the inorganic compound NaCl is a classic example of how a direct electron transfer between atoms occurs by forming ions.
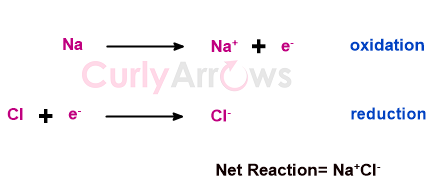
However, only a few organic chemistry reactions are ionic. Therefore, organic chemistry's most oxidation (or reduction) reactions are not direct electron transfers.
The second method of calculating oxidation number assigns a value and a charge to an atom based on the number of electrons it lost or gained, hypothetically behaving as ions.
For example, the oxidation number of Oxygen is -2. The Oxygen atom is devoid of two electrons to reach a stable octet state, and it would always want to attract those two electrons and form a bond with other atoms.
The Hydrogen atom has an oxidation state of +1 since it prefers to donate one electron to another atom and attain a duplet state. Therefore, the assumption that the atom behaves like ions and forms bonds with other atoms is made while calculating oxidation numbers.
Since organic chemistry is mainly concerned with carbon-hydrogen-containing compounds, the second method of calculating the oxidation number for the atom undergoing transformations is cumbersome and misleading and can even generate fractional values.
For example, the average oxidation number of carbon in methane is -4, but in propane, it is -2.67, and in butane, it is -2.5.
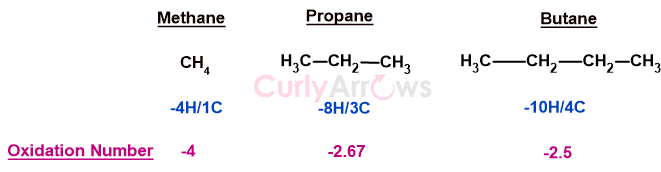
Relying on the oxidation number alone would suggest that butane has a higher oxidation level than propane, though, in fact, they both are hydrocarbons. They have the same oxidation level (alkane level). From an organic chemist’s perspective, they aren’t very different.
(Read about the difference between oxidation state and oxidation level)
To overcome this challenge, instead of assigning the oxidation state to the whole molecule, designating the oxidation state to individual atoms in a molecule based on what is bonded to them would give more useful information.
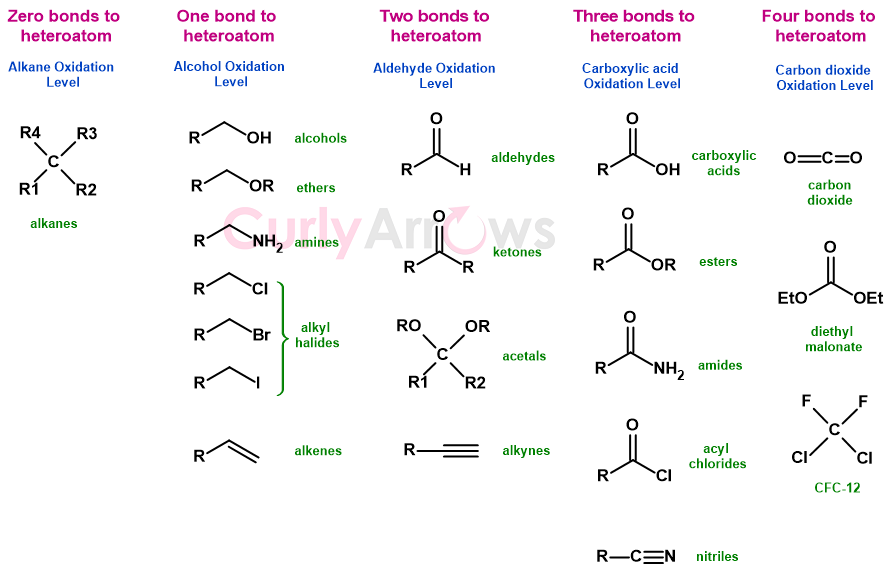
Therefore, organic chemistry takes the carbon atom as a standard, and depending on the heteroatoms (atoms other than C and H) bonded to it, five oxidation levels are categorized since it is the heteroatom that takes away the carbon’s electron density and oxidizes it.
The oxidation levels are:
Alkane (zero bonds to heteroatom)
Alcohol (one bond to heteroatom)
Aldehyde (two bonds to heteroatom)
The carboxylic acid (three bonds to heteroatom)
Carbon dioxide (four bonds to heteroatom)
Here, the number of heteroatoms becomes less critical, but what is essential is the number of bonds to them.
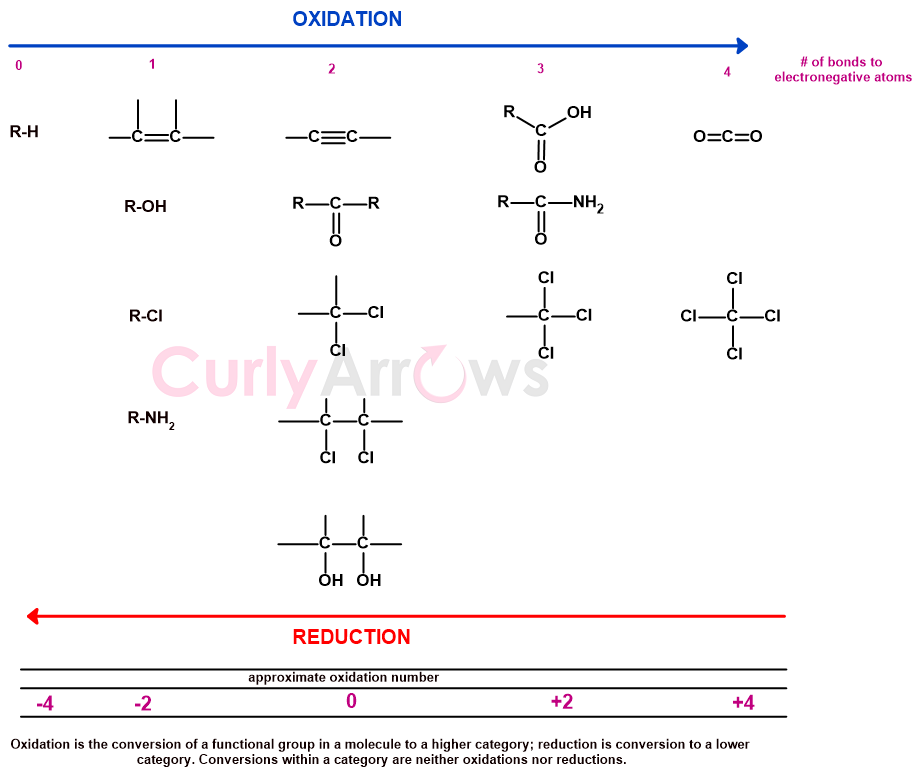
Such changes are seen while a molecule undergoes functional group transformation in chemical reactions. Therefore, oxidation in organic chemistry is a functional group transformation reaction from one category to another where bonds to the heteroatoms increase, and the reduction is the opposite.
The conversion of functional groups in the same category is not oxidation or reduction, as the number of bonds to the heteroatom remains the same.
An exception to this is alkenes and alkynes since they have no heteroatom. However, alkene can be made from alcohols (dehydration) and alkynes from aldehydes (hydration/dehydration) without oxidation or reduction and therefore placed under it.
It is important to remember that all oxidation reactions always occur alongside reductions. That is, there is no oxidation without a concurrent reduction.
The oxidation level of alkane, alcohol, aldehyde, carboxylic acid, and carbon dioxide are then assigned arbitrary oxidation numbers.
The methane (CH4) with no heteroatom attached has the lowest oxidation number, -4, and carbon dioxide (O=C=O) with all four bonds to the heteroatom gets the highest oxidation number, +4.
The methane carbon (CH4) gets -4 because, on comparing carbon with hydrogen, carbon retains all the four bond electrons to itself by a property known as electronegativity. Whereas, in carbon dioxide (CO2), the more electronegative oxygen takes away the four bonds of electron density to itself. As the carbon atom loses four bonds’ electron density, it is assigned an oxidation state of +4.
The remaining oxidation number of -2, 0, and +2 are for one, two, and bonds to the heteroatoms.
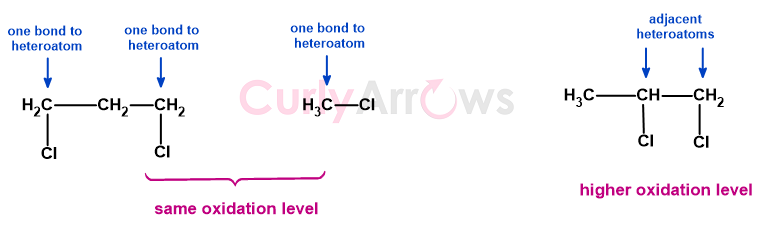
This classification applies to a single carbon atom or two adjacent carbon atoms. A 1,3-dichloropropane has the same oxidation state as chloromethane, but 1,2-dichloropropane is in a higher one.
Reactions enabling Oxidation-Reduction
Organic chemistry tries to understand broadly the reactions that allow such functional group transformations that change the oxidation state and levels.
The direct electron transfer reactions, the addition of electronegative atoms, elimination reactions, substitution reactions where higher electronegative groups are introduced or displaced, and hydrogen atom transfer reactions are some of the mechanistic pathways where oxidation-reduction reactions are encountered.
Oxidation Reactions In Organic Chemistry
An oxidation reaction decreases the number of C-H bonds or increases the number of C-O, C-N, or C-X bonds in a compound (where X is a halogen). Therefore, an oxidation reaction decreases the number of hydrogen and increases the number of oxygen or similar electronegative atoms.
The reactions are classified depending on the type of bond change involved. These groups are -
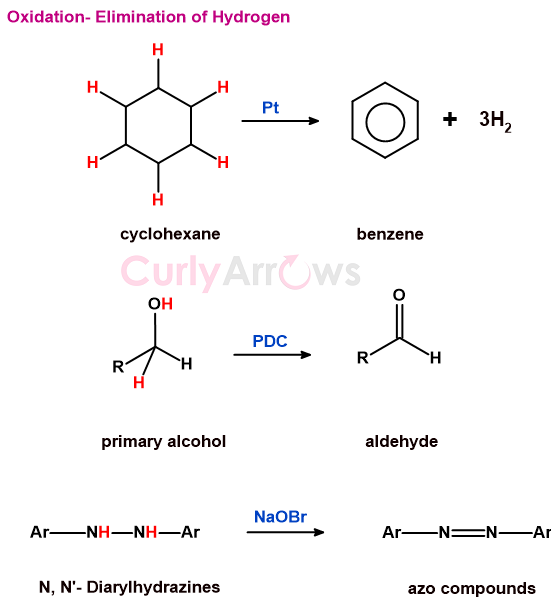
a) eliminations of hydrogen
The decrease in the number of hydrogen atoms is oxidation.
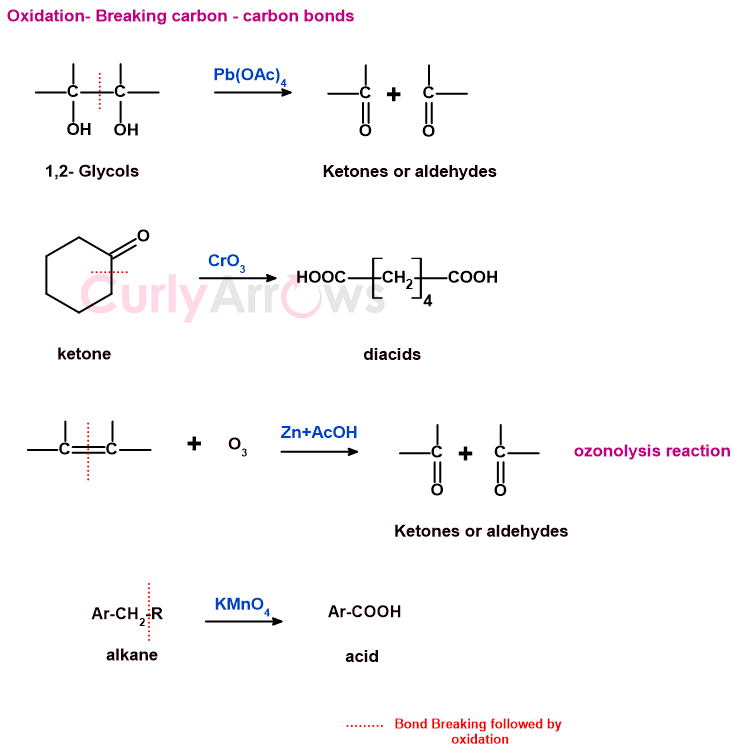
b) reactions involving the breaking of carbon-carbon bonds
In oxidation reactions, the carbon-carbon bonds break, transforming the molecule to a higher oxidation level.
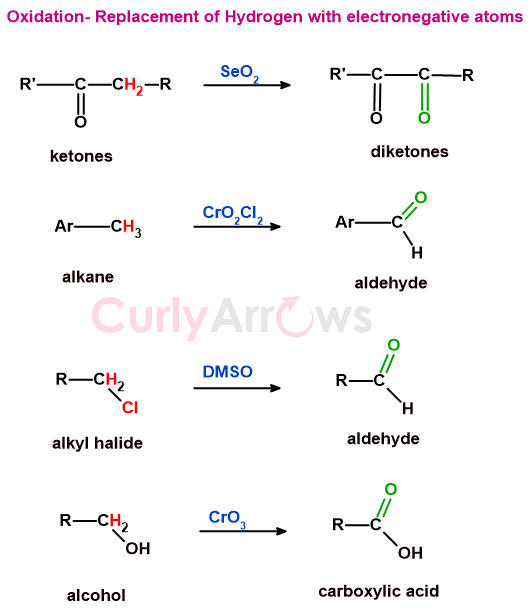
c) reactions involving the replacement of hydrogen by oxygen or other electronegative atoms
Many reactions change the oxidation level by replacing the hydrogen with more electronegative atoms.
d) reactions in which oxygen or an electronegative atom is added to the substrate
Substrates undergo oxidation by adding electronegative atoms to them.
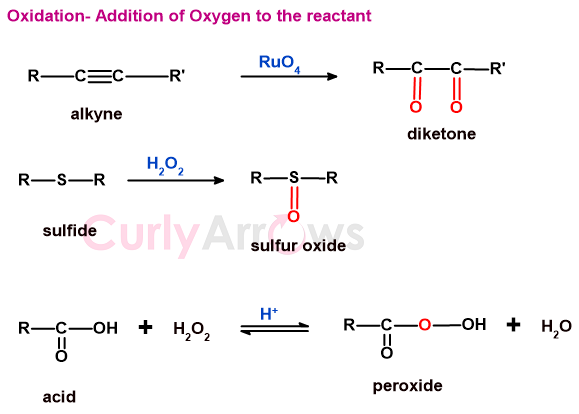
e) oxidative coupling
Oxidative coupling means that two substrate molecules join (couple) by forming a new bond in an oxidation reaction condition.
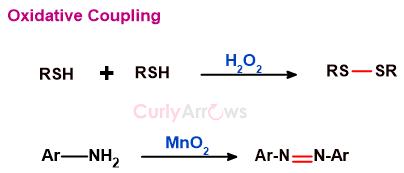
The reagents that enable oxidation are called oxidizing agents.
Reduction Reactions In Organic Chemistry
A reduction reaction increases the number of C-H bonds or decreases the number of C-O, C-N, or C-X bonds in a compound (where X is a halogen). The reactions are classified depending on the type of bond change involved. These groups are -
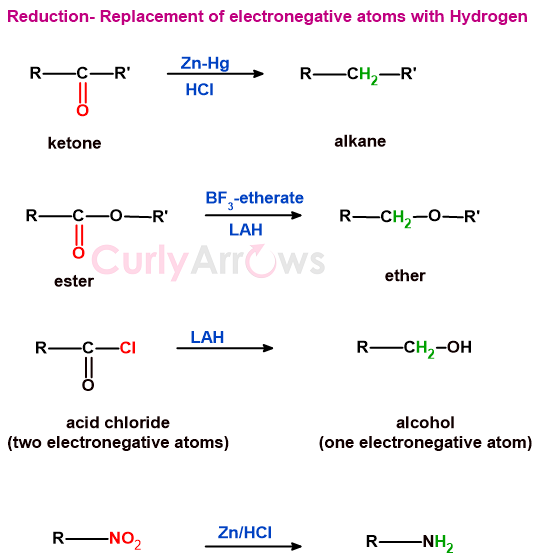
a) reactions involving the replacement of oxygen or other electronegative atoms by hydrogen
The electronegative atoms are replaced with hydrogen in reduction reactions.
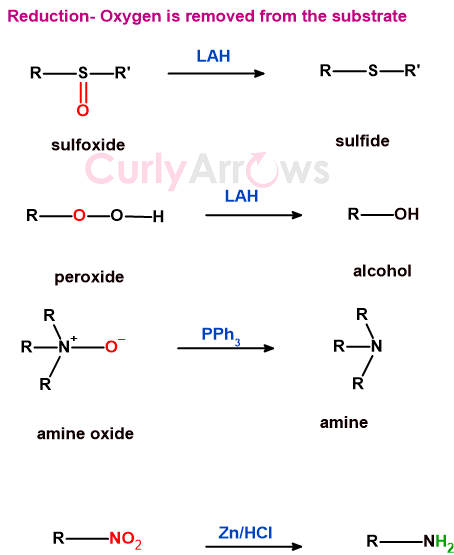
b) reactions in which oxygen or an electronegative atom is removed from the substrate
The reduction reaction removes electronegative atoms from the substrate molecule using reducing agents.
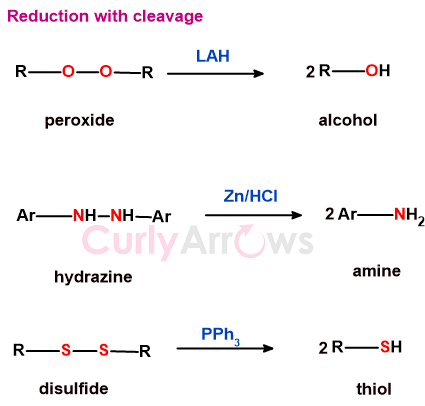
c) reduction with cleavage
Reduction with bond cleavage ensures that the molecule loses at least one electronegative atom.
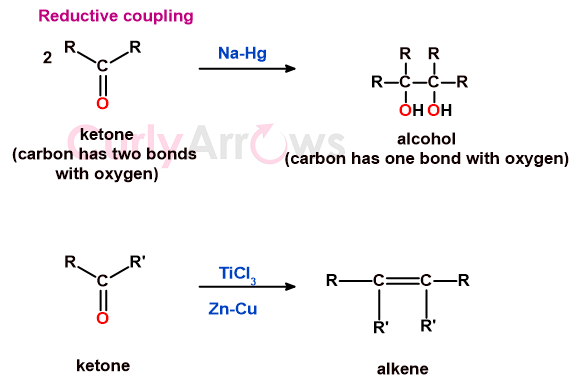
d) reductive coupling
The reductive coupling means that the two substrate molecules under reduction conditions undergo coupling (joining), forming a new covalent bond.
In such reactions, in addition to the removal of electronegative atoms, sometimes a step-down in oxidation level is seen. That is, the substrate with two bonds to the heteroatom is seen with one bond to the heteroatom post-reduction.
The reagents that enable reduction are called reducing agents. Finding a reducing agent that can selectively reduce one group in a molecule without affecting the other is possible.
